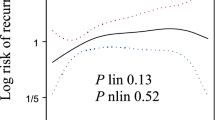
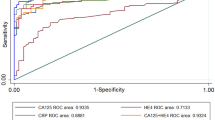
Abstract
The value of the serum tumour marker CA 125 to date has been in the monitoring of ovarian cancer patients for response to therapy and for recurrence of disease. However, despite the availability of serial data on CA 125, the problem of interpreting a change over time is still unsolved. The aim of this study was to assess the ability of CA 125 to monitor patients with ovarian cancer during postoperative chemotherapy. 255 patients with stage IC-IV ovarian cancer were allocated to the tumour marker monitoring study. The evaluation of CA 125 information was based on the analytical imprecision, the normal intra-individual biological variation, the sampling interval, and the cut-off value. Additionally, a new assessment criterion based upon an increment of 2.5 times the baseline CA 125 concentration confirmed by a third measurement was elaborated and the utility investigated. The efficiency of CA 125 for identifying progression and non-progression during first-line chemotherapy was 91.9%. The median lead time for true positive results was 41 days. Using the new elaborated criterion the efficiency of CA 125 for identifying progression and non-progression during first-line chemotherapy was 90.5%. The median lead time for true positive results was 35 days. CA 125 gave reliable prediction of progressive disease during postoperative chemotherapy. The results indicate a high applicability of the presented progression criteria during CA 125 monitoring of patients with changing activity of ovarian cancer. © 2001 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Armitage P and Berry G (1994) Statistical Methods in Medical Research. Blackwell Scientific Publications: Oxford
Bast RC Jr, Klug TL, St John E, Jenison E, Niloff JM, Lazarus H and Berkowitz RS (1983) A radioimmunoassay using a monoclonal antibody to monitor the course of epithelial ovarian cancer. N Engl J Med 309: 883–887
Bast RC Jr, Klug TL, Schaetzl E, Lavin P, Niloff JM, Greber TF, Zurawski VR and Knapp RC (1984) Monitoring human ovarian carcinoma with a combination of CA 125, CA 19–9, and carcinoembryonic antigen. Am J Obstet Gynecol 149: 553–559
Berek JS, Bertelsen K, Du Bois A, Brady MF, Carmichael J, Eisenhauer EA, Gore M, Grenman S, Hamilton TC, Hansen SW, Harper PG, Horvath G, Kaye SB, Lück HJ, Lund B, McGuire WP, Neijt JP, Ozols RF, Parmar M, Piccart-Gebhart MJ, Rosenberg P, Rustin G, Sessa C, Trope C, Tuxen MK, van Rijswijk R, Vergote IB, Vermorken JB and Willemse PHB (1999) Advanced epithelial ovarian cancer: 1998 consensus statements. Ann Oncol 10: S87–S92
Boscato LM and Stuart MC (1988) Heterophilic antibodies: a problem for all immunoassays. Clin Chem 34: 27–33
Browning MCK (1987) Biological variation of the breast-tumour marker CA 15–3: implications for necessary standards of performance and the interpretation of results. Ann Clin Biochem 24: S1-35–S1-36
Browning MCK and McFarlane NP (1990) Objective interpretation of results for tumour markers. J Nucl Med Allied Sci 34: 89–91
Cobas® Core CA 125 II EIA (1996) Roche Diagnostic Systems. Package insert
Collazos J, Genolla J and Ruibal A (1992) CA 125 serum levels in patients with non-neoplastic liver diseases. A clinical and laboratory study. Scand J Clin Lab Invest 52: 201–206
Cruickshank DJ, Terry PB and Fullerton WT (1992) CA 125 -response assessment in epithelial ovarian cancer. Int J Cancer 51: 58–61
Fioretti P, Gadducci A, Ferdeghini M, Bartolini T, Bianchi R and Facchini V (1987) Correlation of CA 125 and CA 19–9 serum levels with clinical course and second-look findings in patients with ovarian carcinoma. Gynecol Oncol 28: 278–283
Fioretti P, Gadducci A, Ferdeghini M, Prontera C, Malagnino G, Facchini V, Marianin G and Bianchi R (1992) The concomitant determination of different serum tumor markers in epithelial ovarian cancer: relevance for monitoring the response to chemotherapy and follow-up of patients. Gynecol Oncol 44: 155–160
Fraser CG and Petersen PH (1991) The importance of imprecision. Ann Clin Biochem 28: 207–211
Fraser CG, Petersen PH and Larsen ML (1990) Setting analytical goals for random analytical error in specific clinical monitoring situations. Clin Chem 36: 1625–1628
Gadducci A, Ferdeghini M, Ceccarini T, Prontera C, Facchini V, Bianchi R and Fioretti P (1990) A comparative evaluation of the ability of serum CA 125, CA 19–9, CA 15–3, CA 50, CA 72–4 and TATI assays in reflecting the course of disease in patients with ovarian carcinoma. Eur J Gynaecol Oncol 11: 127–133
Gadducci A, Ferdeghini M, Rispoli G, Prontera C, Bianchi R and Fioretti P (1991) Comparison of tumor associated trypsin inhibitor (TATI) with CA 125 as a marker for diagnosis and monitoring of epithelial ovarian cancer. Scand J Clin Lab Invest 51: 19–24
Gard GB and Houghton CRS (1994) An assessment of the value of serum CA 125 measurements in the management of epithelial ovarian carcinoma. Gynecol Oncol 53: 283–289
Gion M, Barioli P, Mione R, Cappelli G, Vignati G, Fortunato A, Saracchini S, Biasioli R and Gulisano M (1995) Tumor markers in breast cancer follow-up: A potentially useful parameter still awaiting definitive assessment. Ann Oncol 6: S31–S35
Hising C, Anjegard IM and Einhorn N (1991) Clinical relevance of the CA 125 assay in monitoring of ovarian cancer patients. Am J Clin Oncol 14: 111–114
Krebs HB, Goplerud DR, Kilpatrick SJ, Myers MB and Hunt A (1986) Role of CA 125 as tumor marker in ovarian carcinoma. Obstet Gynecol 67: 473–477
Molina R, Filella X, Bruix J, Mengual P, Bosch J, Calvet X, Jo J and Ballesta AM (1991) Cancer antigen 125 in serum and ascitic fluid of patients with liver diseases. Clin Chem 37: 1379–1383
Nicholson S, Fox M, Epenetos A and Rustin G (1996) Immunoglobulin Inhibiting Reagent®: evaluation of a new method for eliminating spurious elevations in CA 125 caused by HAMA. Int J Biol Markers 11: 46–49
Niloff JM, Knapp RC, Lavin PT, Malkasian GD, Berek JS, Muriel R, Whitney C, Zurawski VR and Bast RC (1986) The CA 125 assay as a predictor of clinical recurrence in epithelial ovarian cancer. Am J Obstet Gynecol 155: 56–60
Panza N, Pacilio G, Campanella L, Peluso G, Battista C, Amoriello A, Utech W, Vacca C and Lombardi G (1988) Cancer antigen 125, tissue polypeptide antigen, carcinoembryonic antigen, and beta-chain human chorionic gonadotropin as serum markers of epithelial ovarian carcinoma. Cancer 61: 76–83
Plebani M, Giacomini A, Beghi L, De Paoli M, Roveroni G, Galeotti F, Corsini A and Fraser CG (1996) Serum tumor markers in monitoring patients: interpretation of results using analytical and biological variation. Anticancer Res 16: 2249–2252
Ruibal A, Encabo G, Martinez-Miralles E, Murcia C, Capdevila JA, Salgado A and Martinez-Vasquez JM (1984) CA 125 seric levels in non malignant pathologies. Bull Cancer 71: 145–146
Rustin GJS, Nelstrop A, Stilwell J and Lambert HE (1992) Savings obtained by CA-125 measurements during therapy for ovarian carcinoma. Eur J Cancer 28: 79–82
Rustin GJS, van der Burg MEL and Berek JS (1993) Tumour markers. Ann Oncol 4: S71–S77
Rustin GJS, Nelstrop AE, Tuxen MK and Lambert HE (1996) Defining progression of ovarian carcinoma during follow-up according to CA 125: A North Thames Ovary Group study. Ann Oncol 7: 361–364
Rustin GJ, Nelstrop AE, Crawford M, Ledermann J, Lambert HE, Coleman Johnson J, Evans H, Brown S and Oster W (1997) Phase II trial of oral altretamine for relapsed ovarian carcinoma: evaluation of defining response by serum CA125. J Clin Oncol 15: 172–176
Storm HH, Pihl J, Michelsen E and Nielsen AL (1996) Cancer incidence in Denmark 1993. Danish Cancer Society
Sölétormos G, Nielsen D, Schiøler V, Skovsgaard T, Winkel P, Mouridsen HT and Dombernowsky P (1993a) A novel method for monitoring high-risk breast cancer with tumor markers: CA 15.3 compared to CEA and TPA. Ann Oncol 4: 861–869
Sölétormos G, Schiøler V, Nielsen D, Skovsgaard T and Dombernowsky P (1993b) Interpretation of results for tumor markers on the basis of analytical imprecision and biological variation. Clin Chem 39: 2077–2083
Sölétormos G, Nielsen D, Schiøler V, Skovsgaard T and Dombernowsky P (1996) Tumor markers cancer antigen 15.3, carcinoembryonic antigen, and tissue polypeptide antigen for monitoring metastatic breast cancer during first-line chemotherapy and follow-up. Clin Chem 42: 564–575
Thigpen T, Vance RB, McGuire WP, Hoskins WJ and Brady M (1995) The role of paclitaxel in the management of coelomic epithelial carcinoma of the ovary: a review with emphasis on the Gynecologic Oncology Group experience. Semin Oncol 22: 23–31
Tuxen MK, Sölétormos G and Dombernowsky P (1995) Tumor markers in the management of patients with ovarian cancer. Cancer Treat Rev 21: 215–245
Tuxen MK, Sölétormos G, Rustin GJS, Nelstrop AE and Dombernowsky P (2000) Biological variation and analytical imprecision of CA 125 in patients with ovarian cancer. Scand J Clin Lab Invest 60: 713-722
van der Burg ME, Lammes FB and Verweij J (1990) The role of CA 125 in the early diagnosis of progressive disease in ovarian cancer. Ann Oncol 1: 301–302
Vergote IB, Abeler VM, Bormer OP, Stigbrand T, Trope C and Nustad K (1992) CA 125 and placental alkaline phosphatase as serum tumor markers in epithelial ovarian carcinoma. Tumor Biol 13: 168–174
Ward BG, McGuckin MA, Ramm LE, Coglan M, Sanderson B, Tripcony L and Free KE (1993) The management of ovarian carcinoma is improved by the use of cancer-associated serum antigen and CA 125 assays. Cancer 71: 430–438
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Tuxen, M., Sölétormos, G. & Dombernowsky, P. Serum tumour marker CA 125 in monitoring of ovarian cancer during first-line chemotherapy. Br J Cancer 84, 1301–1307 (2001). https://doi.org/10.1054/bjoc.2001.1787
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.1787
Keywords
This article is cited by
-
Peritoneal tuberculosis mimicking carcinomatous ascites in a child living in a low prevalence country: a case report
Italian Journal of Pediatrics (2020)
-
Circulating tumor cells as a new and additional approach to follow-up patients with serous low-grade ovarian adenocarcinoma – a case report and review of the literature
Applied Cancer Research (2017)
-
Prediction of 5-year survival in advanced-stage ovarian cancer patients based on computed tomography peritoneal carcinomatosis index
Abdominal Radiology (2016)
-
A repertoire of biomarkers helps in detection and assessment of therapeutic response in epithelial ovarian cancer
Molecular and Cellular Biochemistry (2014)
-
Dynamic optimal strategy for monitoring disease recurrence
Science China Mathematics (2012)