Abstract
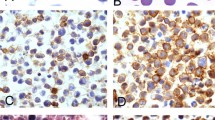
We have examined expression of the Epstein–Barr virus (EBV) latent membrane protein-1 (LMP1) in the malignant Hodgkin and Reed–Sternberg (HRS) cells of Hodgkin's disease (HD) and its impact on response to treatment and on survival. Paraffin tissue from 100 adult immunocompetent patients with HD were analysed using immunohistochemistry to identify LMP1 expression. According to the Rye classification, 8% of patients had lymphocyte predominance (LP) subtype, 48% had nodular sclerosis (NS) disease, 37% were of the mixed cellularity (MC) subtype and 7% were of the lymphocyte depletion (LD) subtype. During the five year follow-up period 27 patients died and 74 patients achieved a complete remission. Patients with LD subtype were older (P = 0.03), less frequently achieved complete remission (P = 0.01), had shorter disease-free survival (P = 0.01) and overall survival (P = 0.002) compared with the other subtypes of HD. LMP1 expression was found in the tumour cells of 26% of cases of HD. LMP1 expression was less common in NS disease than in the other subtypes (P = 0.05), whereas an association between MC subtype and LMP1 expression was not found (P = 0.22). Using the log-rank test there were no differences in overall survival or disease-free survival based on EBV status either when all patients were analysed or when LD and LP subtypes were excluded. However, the presence of EBV was associated with significantly longer disease-free survival in the subgroup of patients ≤ 30 years old (P = 0.02) and in those patients ≤ 34 years old (P = 0.05). In contrast, there was a trend for shorter disease-free survival for EBV-positive patients in the subgroup > 35 years old, but this difference was not statistically significant (P = 0.11). A similar trend was observed in patients > 50 years old. Analysis of the impact of LMP1 expression in patients who had different stage and B symptoms status showed that expression of EBV was associated with longer disease-free survival (P = 0.019) in early stage (1 + 2) patients without B symptoms. Significant differences in the other subgroups based on EBV status was not found. Factors adversely affecting the likelihood to achieve a complete remission were: absence of LMP1 expression (OR 6.4, 95% Cl 1.07–38.5, P = 0.04), age (OR 1.68, 95%Cl 1.15–2.5, P = 0.007) and subtype of HD (OR 3.32, 95%Cl 1.11–9.94, P = 0.03). Age and subtype of HD had an independent impact on overall survival (P = 0.01). We conclude that expression of LMP1 in HRS cells has a favourable impact on prognosis for HD patients, but that this effect may be restricted to young adult patients and those with early stage disease. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Axdorph U, Porwit-MacDonald A, Sjoberg J, Grimfors G, Ekman M, Wang W, Biberfeld P and Bjorkholm M (1999) Epstein-Barr virus expression in Hodgkin's disease in relation to patient characteristics, serum factors and blood lymphocyte function. Br J Cancer 81: 1182–1187
Clarke CA, Glaser SL, Dorfman RF, Mann RB, DiGiuseppe JA, Prehn A and Amninder RF (2000) Epstein-Barr virus and survival after Hodgkin's disease in a population-based series of women. International Association for Research on Epstein–Barr virus and Associated diseases Ninth Biennial Conference. 22–27th June, Yale University: New Haven, USA
Claviez A, Tiemann M, Peters J, Kreipe H, Schneppenheim R and Parwaresch R (1994) The impact of EBV, proliferation rate, and Bcl-2 expression in Hodgkin's disease in childhood. Ann Hematol 68: 61–66
Coates PJ, Mak WP, Slavin G and d'Ardenne AJ (1991) Detection of single copies of Epstein–Barr virus in paraffin wax sections by non-radioactive in situ hybridisation. J Clin Pathol 44: 487–491
Cossman J, Messineo C and Bagg A (1998) Reed-Stemberg cell: survival in a hostile see. Lab Invest 78: 229–235
Dukers DF, Jaspars LH, Vos W, Oudejans JJ, Hayes D, Cillessen S, Middeldorp JM and Meijer CJLM (2000) Quantitative immunohistochemical analysis of cytokine profiles in Epstein-Barr virus-positive and -negative cases of Hodgkin's disease. J Pathol 190: 143–149
Enblad G, Sandvej K, Lennette E, Sundstrom C, Klein G, Glimelius B and Pallesen G (1997) Lack of correlation between EBV serology and presence of EBV in the Hodgkin and Reed-Sternberg cells of patients with Hodgkin's disease. Int J Cancer 72: 394–397
Engel M, Essop MF, Close P, Hartley P, Pallesen G and Sinclair-Smith C (2000) Improved prognosis of Epstein-Barr virus associated childhood Hodgkin's lymphoma: study of 47 South African cases. J clin Pathol 53: 182–186
Fellbaum C, Hansmann M-L, Niedermeyer H, Kraus I, Alavaikko MJ, Busch R, Putz B, Fischer R and Hofler H (1992) Influence of Epstein-Barr virus genomes on patient survival in Hodgkin's disease. Am J Clin Pathol 98: 319–323
Frisan T, Sjoberg J, Dolcetti R, Boiocchi M, De Re V, Carbone A, Brautbar C, Battat S, Biberfeld P and Eckman M (1995) Local suppression of Epstein-Barr virus (EBV)-specific cytotoxicity in biopsies of EBV-positive Hodgkin's disease. Blood 86: 1493–1501
Glaser SL and Jarrett RF The epidemiology of Hodgkin's disease (1996). Baillieres Clin Haematol 9: 01–16
Glaser SL, Lin RJ, Stewart SL, Ambinder RF, Jarrett RF, Brousset P, Pallesen G, Gulley ML, Khan G, O'Grady J, Hummel M, Preciado MV, Knecht H, Chan JK and Claviez A (1997) Epstein-Barr virus-associated Hodgkin's disease: epidemiologic characteristics in international data. Int J Cancer 70: 375–382
Glickman JN, Howe JG and Steitz JA (1988) Structural analysis of EBER1 and EBER2 ribonucleoprotein particles present in Epstein-Barr virus infected cells. J Virol 62: 902–911
Gruss H-J, Pinto A, Dystler J, Poppema S and Herrmann F (1997) Hodgkin's disease: a tumor with disturbed immunological pathways. Immunol Today 18: 156–163
Gutensohn N and Cole P (1980) Epidemiology of Hodgkin's disease. Semin Oncol 7: 92–102
Henderson S, Rowe M, Gregory C, Croom-Carter D, Wang F, Longnecker R, Kieff E and Rickinson A (1991) Induction of bcl-2 expression by Epstein-Barr virus latent membrane protein 1 protects infected B cells from programmed cell death. Cell 65: 1107–1115
Herbst H, Steinbecker E, Niedobitek G, Young LS, Brooks L, Muller-Lantzsch N and Stein H (1992). 80
Herbst H, Foss HD, Samol J, Araujo I, Klotzbach H, Krause H, Agathanggelou A, Niedobitek G and Stein H (1996) Frequent expression of interleukin-10 by Epstein-Barr virus-harboring tumor cells of Hodgkin's disease. Blood 87: 2918–2929
Hummel M, Anagnostopoulos I, Dallenbach F, Korbjuhn P, Dimmler C and Stein H (1992) EBV infection patterns in Hodgkin's disease and normal lymphoid tissue: expression and cellular localization of gene products. Brit J Haematol 82: 689–694
Hummel M, Ziemann K, Lammert H, Pileri S, Sabattini E and Stein H (1995) Hodgkin's disease with monoclonal and polyclonal populations of Reed-Sternberg cells. N Engl J Med 333: 901–906
Jiwa NM, Oudejans JJ, Bai MC, van den Brule AJ, Horstman A, Vos A, van der Valk P, Kluin PM, Walboomers JM and Meijer CJ (1995) Expression of bcl-2 protein and transcription of the Epstein-Barr virus homologue BHRF-1 in Hodgkin's disease: implications for different pathogenic mechanisms. Histopathology 26: 547–553
Kanzler H, Kuppers R, Hansmann ML and Rajewsky K (1996) Hodgkin and Reed-Sternberg cells in Hodgkin's disease represent the outgrowth of a dominant tumor clone derived from (crippled) germinal center B cells. J Exp Med 184: 1495–1505
Kaufman D and Longo DL Hodgkin's disease. (1992). Crit Rev Oncol Hematol 13: 135–187
Khanna R, Burrows SR, Nicholls J and Poulsen LM (1998) Identification of cytotoxic T cell epitopes within Epstein-Barr virus (EBV) oncogene latent membrane protein 1 (LMP1): evidence for HLA A2 supertype-restricted immune recognition of EBV-infected cells by LMP1-specific cytotoxic T lymphocytes. Eur J Immunol 28: 451–458
Kordek R, Jesionek-Kupnicka D, Biernat W and Wozniak L (1996) Expression of the latent membrane protein of Epstein-Barr virus in Hodgkin's disease. Age and subtype distribution in Polish patients. Acta Haematol Pol 27: 15–20
Lee SP, Constandinou CM, Thomas WA, Croom-Carter D, Blake NW, Murray PG, Crocker J and Rickinson AB (1998) Antigen presenting phenotype of Hodgkin Reed-Sternberg cells: analysis of the HLA class I processing pathway and the effects of interleukin-10 on Epstein-Barr virus-specific cytotoxic T-cell recognition. Blood 92: 1020–1030
Levine PH, Ablashi DV, Berard CW, Carbone PP, Waggoner DE and Malan L (1971) Elevated antibody titers to Epstein-Barr virus in Hodgkin's disease. Cancer 27: 416–421
Macak J, Habanec B and Fabian P (2000) Detection of Epstein-Barr virus in Hodgkin's lymphoma. Neoplasma 47: 156–161
MacMahon B Epidemiology of Hodgkin's disease (1966). Cancer Res 26: 1189–1201
Morente MM, Piris MA, Abraira V, Acevedo A, Aguilera B, Bellas C, Fraga M, Garcia-Del-Moral R, Gomez-Marcos F, Menarguez J, Oliva H, Sanchez-Beato M and Montalban C (1997) Adverse clinical outcome in Hodgkin's disease is associated with loss of retinoblastoma protein expression, high Ki67 proliferation index, and absence of Epstein-Barr virus-latent membrane protein 1 expression. Blood 90: 2429–2436
Mueller N, Evans A, Harris NL, Cornstock GW, Jellum E, Magnus K, Orentreich N, Polk BF and Vogelman J (1989) Hodgkin's disease and Epstein-Barr virus. Altered antibody pattern before diagnosis. N Engl J Med 32: 689–695
Murray PG, Billingham LJ, Hassan HT, Flavell JR, Nelson PN, Scott K, Reynolds G, Constandinou CM, Kerr DJ, Devey EC, Crocker J and Young LS (1999) Effect of Epstein-Barr virus infection on response to chemotherapy and survival in Hodgkin's disease. Blood 94: 442–447
Murray PG, Constandinou CM, Crocker J, Young LS and Ambinder RF (1998) Analysis of major histocompatibility complex class I, TAP expression, and LMP2 epitope sequence in Epstein-Barr virus-positive Hodgkin's disease. Blood 92: 2477–2483
Murray PG, Young LS, Rowe M and Crocker J (1992) Immunohistochemical demonstration of the Epstein-Barr virus encoded latent membrane protein in paraffin sections of Hodgkin's disease. J Pathol 166: 1–5
Nakagomi H, Dolcetti R, Bejarano MT, Pisa P, Kiessling R and Masucci MG (1994) The Epstein-Barr virus latent membrane protein-1 (LMP1) induces interleukin-10 production in Burkitt lymphoma lines. Int J Cancer 57: 240–244
Naresh KN, Johnson J, Srinivas V, Soman CS, Saikia T, Advani SH, Badwe RA, Dinshaw KA, Muckaden M, Magrath I and Bhatia K (2000) Epstein-Barr virus association in classical Hodgkin's disease provides survival advantage to patients and correlates with higher expression of proliferation markers in Reed-Sternberg cells. Ann Oncol 11: 91–96
Niedobitek G, Kremmer E, Herbst H, Whitehead L, Dawson CW, Niedobitek E, von Ostau C, Rooney N, Grässer FA and Young LS (1997) Immunohistochemical Detection of the Epstein-Barr Virus – Encoded Latent Membrane Protein 2A in Hodgkin's Disease and Infectious Mononucleosis. Blood 90: 1664–1672
Oudejans JJ, Jiwa NM, Kummer JA, Horstman A, Vos W, Baak JP, Kluin PM, van der Valk P, Walboomers JM and Meijer CJLM (1996) Analysis of major histocompatibility complex class I expression on Reed-Sternberg cells in relation to the cytotoxic T-cell response in Epstein-Barr virus-positive and -negative Hodgkin's disease. Blood 87: 3844–3851
Oudejans JJ, Jiwa NM and Meijer CJ (1997) Epstein-Barr virus in Hodgkin's disease: more than just an innocent bystander. J Pathol 181: 353–356
Pallesen G, Hamilton-Dutoit SJ, Rowe M and Young LS (1991) Expression of Epstein-Barr virus latent gene products in tumour cells of Hodgkin's disease. Lancet 337: 320–322
Roskrow MA, Suzuki N, Gan YJ, Sixbey JW, Ng CY, Kimbrough S, Hudson M, Brenner MK, Heslop HE and Rooney CM (1998) Epstein-Barr virus (EBV)-specific cytotoxic T lymphocytes for the treatment of patients with EBV-positive relapsed Hodgkin's disease. Blood 91: 2925–2934
Shibata D, Hansmann M-L, Weiss LM and Nathwani BN (1991) Epstein-Barr virus infections and Hodgkin's disease: a study of fixed tissue using the polymerase chain reaction. Hum Pathol 22: 1262–1267
Sing A, Ambinder RF, Hokg DJ, Jensen M, Batten V, Petersdorf E and Greenberg PD (1997) Isolation of Epstein-Barr virus (EBV)-specific cytotoxic T-lymphocytes that lyse Reed-Sternberg cells: Implications for immune-mediated therapy for Hodgkin's disease. Blood 89: 1978–1986
Vestlev PM, Pallesen G, Sandvej K, Hamilton-Dutoit SJ and Bendtzen SM (1992) Prognosis of Hodgkin's disease is not influenced by Epstein-Barr virus latent membrane protein. Int J Cancer 50: 670–671
Wang D, Liebowitz D and Kieff E (1985) An EBV membrane protein expressed in immortalized lymphocytes transforms established rodent cells. Cell 43: 831–840
Wang S, Rowe M and Lundgren E (1996) Expression of the Epstein Barr virus transforming protein LMP1 causes a rapid and transient stimulation of the Bcl-2 homologue Mcl-1 levels in B-cell lines. Cancer Res 56: 4610–4613
Weiss LM, Strickler JG, Warnke JA, Purtilo DT and Sklar J (1987) Epstein-Barr viral DNA in tissues of Hodgkin's disease. Am J Pathol 129: 86–91
Wu TC, Mann RB, Charache P, Hayward SD, Staal S, Lambe BC and Ambinder RF (1990) Detection of EBV gene expression in Reed-Sternberg cells of Hodgkin's disease. Int J Cancer 46: 801–804
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Glavina-Durdov, M., Jakic-Razumovic, J., Capkun, V. et al. Assessment of the prognostic impact of the Epstein–Barr virus-encoded latent membrane protein-1 expression in Hodgkin's disease. Br J Cancer 84, 1227–1234 (2001). https://doi.org/10.1054/bjoc.2001.1774
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.1774
This article is cited by
-
Impact of the Epstein–Barr virus positivity on Hodgkin's lymphoma in a large cohort from a single institute in Korea
Annals of Hematology (2012)
-
The effect of Epstein–Barr virus status on clinical outcome in Hodgkin’s lymphoma
Annals of Hematology (2006)
-
Constitutive activation of STAT3 and STAT5 is present in the majority of nasopharyngeal carcinoma and correlates with better prognosis
British Journal of Cancer (2003)
-
Epstein–Barr virus and oncogenesis: from latent genes to tumours
Oncogene (2003)