Abstract
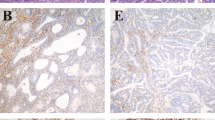
To clarify the possible role of aberrant β-catenin expression during endometrial tumorigenesis, a total of 199 cases of endometrial carcinomas (endometrioid type), as well as 37 cases of simple/complex and 32 of atypical hyperplasias, was consecutively investigated for immunohistochemistry, along with 141 normal endometrial samples distant from carcinomas. Of 199 carcinoma cases, 73 tumours as well as 44 normal samples were also analysed using a combination of RT-PCR and Southern blot hybridization, Western blot, and mutation gene assays. Cell membrane β-catenin immunoreactivity showed a stepwise decrease from normal, through atypical hyperplasia, to grade 3 carcinomas. In contrast, the nuclear accumulation in atypical hyperplasias and grade 1 or 2 tumours was higher than in simple/complex hyperplasias. Mutations in exon 3 of the β-catenin gene involving codons 33, 34, 37, 41, and 45 were observed in 16 (22.9%) of 70 endometrial carcinomas, as well as 3 (12.5%) of 24 atypical hyperplasias, the results being significantly related to low membrane and high nuclear immunoreactivity but not relative mRNA expression levels, suggesting that the gene mutations may be closely associated with changes in subcellular distribution. In addition to significant association between β-catenin mutation and low grade histological malignancy (P=0.048), the mutations were detected in none of 15 and 13 (26%) of 50 tumours with or without lymph node metastasis, the difference being significant (P=0.027). These findings suggest that β-catenin abnormalities may play an important role in a relatively early event during the endometrial hyperplasia-carcinoma sequence. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Aberle H, Bauer A, Stappert J, Kispert A and Kemler R (1997) β-catenin is a target for the ubiquitin-proteasome pathway. EMBO J 16: 3797–3804
Beherens J, Kries JPV, Kuhl U, Bruhn L, Wedlich D, Grosschedl R and Birchmeier W (1996) Functional interaction of β-catenin with the transcription factor LEF-1. Nature 382: 638–642
Berchuck A and Boyd J (1995) Molecular basis of endometrial cancer. Cancer 76: 2034–2040
de Boer CJ, van Dorst E, van Krieken H, Jansen-van Rhiji CM, Warnaar SO, Fleuren GJ and Litvinov SV (1999) Changing roles of cadherins and catenins during progression of squamous intraepithelial lesions in the uterine cervix. Am J Pathol 155: 505–515
Deligdisch L and Cohen CJ (1985) Histologic correlates and virulence implications of endometrial carcinoma associated with adenomatous hyperplasia. Cancer 56: 1452–1455
Fujimoto J, Ichigo S, Hori M, Morishita S and Tamaya T (1996) Progestins and danazol effect on cell-to-cell adhesion, and E-cadherin and α- and β-catenin mRNA expressions. J Steroid Biochem Mol Biol 57: 275–282
Fukuchi T, Sakamoto M, Tsuda H, Maruyama K, Nozawa S and Hirohashi S (1998) β-catenin mutation in carcinoma of the uterine endometrium. Cancer Res 58: 3526–3528
Garcia-Rostan G, Tallini G, Herrero A, D′Aquila TG, Carcangiu ML and Rimm DL (1999) Frequent mutation and nuclear localization of β-catenin in anaplastic thyroid carcinoma. Cancer Res 59: 1811–1815
Gumbiner BM (1996) Cell adhesion: the molecular basis of tissue architecture and morphogenesis. Cell 84: 345–357
Hao XP, Tomlinson I, Ilyas M, Palazzo JP and Talbot IC (1997) Reciprocity between membranous and nuclear expression of β-catenin in colorectal tumours. Virchows Archiv 431: 167–172
Hinck L, Nelson WJ and Papkoff JJ (1994) Wnt-1 modulates cell-cell adhesion in mammalian cells by stabilizing β-catenin binding to the cell adhesion protein cadherin. J Cell Biol 124: 729–741
Horstmann MA, Posl M, Scholz RB, Anderegg B, Simon P, Baumgaertl K, Delling G and Kabisch H (1997) Frequent reduction or loss of DCC gene expression in human osteosarcoma. Br J Cancer 75: 1309–1317
Ilyas M, Tomlison IPM, Rowan A, Pignatelli M and Bodmer WF (1997) β-catenin mutations in cell lines established from human colorectal cancers. Proc Natl Acad Sci USA 94: 10330–10334
Iwao K, Nakamori S, Kameyama M, Imaoka S, Kinoshita M, Fukui T, Ishiguro S, Nakamura Y and Miyoshi Y (1998) Activation of the β-catenin gene by interstitial deletions involving exon 3 in primary colorectal carcinomas without adenomatous polyposis coli mutations. Cancer Res 58: 1021–1026
Jones MH, Koi S, Fujimoto I, Hasumi K, Kato K and Nakamura Y (1994) Allelotype of uterine carcinoma by analysis of RFLP and microsatellite polymorphisms: frequent loss of heterozygosity on chromosome arms 3p, 9q, 10q, and 17p. Cenes Chromosomes Cancer 9: 119–123
Kimura Y, Shiozaki H, Doki Y, Yamamoto M, Utsunomiya T, Kawanishi K, Fukuchi N, Inoue M, Tsujinaka T and Monden M (1999) Cytoplasmic β-catenin in esophageal cancers. Int J Cancer 84: 174–178
Kobayashi K, Sagae S, Nishioka Y, Tokino T and Kudo R (1999) Mutations of the β-catenin gene in endometrial carcinomas. Jpn J Cancer Res 90: 55–59
Kondo Y, Kanai Y, Sakamoto M, Genda T, Mizokami M, Ueda R and Hirohashi S (1999) β-catenin accumulation and mutation of exon 3 of the β-catenin gene in hepatocellular carcinoma. Jpn J Cancer Res 90: 1301–1309
Kurman RJ, Kaminski PF and Norris HJ (1985) The behavior of endometrial hyperplasia: a long-term study of “untreated” hyperplasia in 170 patients. Cancer 56: 403–412
Mirabelli-Primdahl L, Gryfe R, Kim H, Millar A, Luceri C, Dale D, Holowaty E, Bapat B, Gallinger S and Redston M (1999) β-catenin mutations are specific for colorectal carcinomas with microsatellite instability but occur in endometrial carcinomas irrespective of mutator pathway. Cancer Res 59: 3346–3351
Molenaar M, Wetering MVD, Oosterwegel M, Peterson-Maduro J, Godsave S, Korinek V, Roose J, Destree O and Clevers H (1996) XTcf-3 transcription factor mediates β-catenin-induced axis formation in xenopus embryos. Cell 86: 391–399
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B and Kinzler KW (1997) Activation of β-catenin-Tcf signaling in colon cancer by mutations in β-catenin or APC. Science 275: 1787–1790
Nhieu JTV, Renard CA, Wei Y, Cherqui D, Zafrani ES and Buendia MA (1999) Nuclear accumulation of mutated-catenin in hepatocellular carcinoma is associated with increased cell proliferation. Am J Pathol 155: 703–710
Orford K, Crockett C, Jensen JP, Weissman AM and Byers SW (1997) Serine phosphorylation-regulated ubiquitination and degradation of β-catenin. J Biol Chem 272: 24735–24738
Ozawa M, Baribault H and Kemler R (1989) The cytoplasmic domain of the cell adhesion molecule uvomorulin associates with three independent proteins structurally related in different species. EMBO J 8: 1711–1718
Palacios J and Gamallo C (1998) Mutations in the β-catenin gene (CTNNB1) in endometrioid ovarian carcinomas. Cancer Res 58: 1344–1347
Peifer M, McCrea PD, Green KJ, Wieschaus E and Gumbiner BM (1992) The vertebrate adhesive junction protein β-catenin and plakoglobin and the Drosophila segment polarity gene armadillo form a multigene family with similar properties. J Cell Biol 118: 681–691
Rubinfeld B, Robbins P, El-Gamli M, Albert I, Porfiri E and Polakis P (1997) Stabilization of β-catenin by genetic defects in melanoma cell lines. Science 275: 1790–1792
Saegusa M and Okayasu I (1997) Down-regulation of bcl-2 expression is closely related to squamous differentiation and progesterone therapy in endometrial carcinomas. J Pathol 182: 429–436
Saegusa M, Hashimura M, Hara A and Okayasu I (1999) Loss of expression of the gene deleted in colon carcinoma (DCC) is closely related to histologic differentiation and lymph node metastasis in endometrial carcinoma. Cancer 85: 453–464
Samowitz WS, Powers MD, Spirio LN, Nollet F, van Roy F and Slattery ML (1999) β-catenin mutations are more frequent in small colorectal adenomas than in larger adenomas and invasive carcinomas. Cancer Res 59: 1442–1444
Sparks AB, Morin PJ, Vogelstein B and Kinzler KW (1998) Mutational analysis of the APC/β-catenin/Tcf pathway in colorectal cancer. Cancer Res 58: 1130–1134
Woodman AC, Sugiyama M, Yoshida K, Sugino T, Borgya A, Goodison S, Matsumura Y and Tarin D (1996) Analysis of anomalous CD44 gene expression in human breast, bladder, and colon cancer and correlation of observed mRNA and protein isoforms. Am J Pathol 149: 1519–1530
Yoshinaga K, Sasano H, Furukawa T, Yamakawa H, Yuki M, Sato S, Yajima A and Horii A (1998) The PTEN, BAX, and IGFIIR genes are mutated in endometrial atypical hyperplasia. Jpn J Cancer Res 89: 985–990
Zaino RJ, Kurman R, Herbold D, Gliedman J, Bundy BN, Voet R and Advani H (1991) The significance of squamous differentiation in endometrial carcinoma. Cancer 68: 2293–2302
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Saegusa, M., Hashimura, M., Yoshida, T. et al. β- Catenin mutations and aberrant nuclear expression during endometrial tumorigenesis. Br J Cancer 84, 209–217 (2001). https://doi.org/10.1054/bjoc.2000.1581
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1581
Keywords
This article is cited by
-
Type-II endometrial cancer: role of adipokines
Archives of Gynecology and Obstetrics (2019)
-
Molecular expression characteristics confirm the malignancy concealed by morphological alterations in endometrial cancer after fertility-preserving treatment
Archives of Gynecology and Obstetrics (2019)
-
CTNNB1 (beta-catenin) mutation identifies low grade, early stage endometrial cancer patients at increased risk of recurrence
Modern Pathology (2017)