Abstract
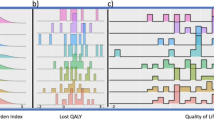
Health Related Quality of Life (HRQoL) instruments are increasingly important in evaluating health care, especially in cancer trials. When planning a trial, one essential step is the calculation of a sample size, which will allow a reasonable chance (power) of detecting a pre-specified difference (effect size) at a given level of statistical significance. It is almost mandatory to include this calculation in research protocols. Many researchers quote means and standard deviations to determine effect sizes, and assume the data will have a Normal distribution to calculate their required sample size. We have investigated the distribution of scores for two commonly used HRQoL instruments completed by lung cancer patients, and have established that scores do not have the Normal distribution form. We demonstrate that an assumption of Normality can lead to unrealistically sized studies. Our recommendation is to use a technique that is based on the fact that the HRQoL data are ordinal and makes minimal but realistic assumptions. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Campbell MJ, Julious SA and Altman DG (1995) Estimating sample size for binary, ordered categorical, and continuous outcomes in two group comparisons. Br Med J 311: 1145–1148
Campbell MJ, Julious SA and George SL (1996) Estimating sample sizes for studies using the SF-36 health survey (reply to letter). Epidemiol Comm Health 50: 473–474
Campbell MJ, Walker SJ, George SL, Machin D and Julious SA (2000) A review of the use of the main quality of life measures, and sample size determination for quality of life measures, particularly in cancer trials. In: Advanced Handbook in Evidence Based Healthcare, Steven A, Abrams KR, Brazier J, Fitzpatrick R, Lilford RJ (eds) Sage Publications: London
de Haes JCJM and van Knippenberg FCE (1985) The quality of life of cancer patients – a review of the literature. Soc Sc Med 20: 809–817
de Haes JCJM, van Knippenberg FCE and Neijt JP (1990) Measuring psychological and physical distress in cancer patients: structure and application of the Rotterdam Symptom Checklist. Br J Cancer 62: 1034–1038
Fayers PM and Machin D (1995) Sample size – how many patients are necessary?. Br J Cancer 72: 1–9
Fayers PM and Machin D (2000) Quality of Life: Assessment, Analysis and Interpretation. John Wiley: Chichester
Julious SA and Campbell MJ (1996) Sample size calculations for ordered categorical data. Stats in Med 15: 1065–1066
Julious SA, George S and Campbell MJ (1995) Sample size for studies using the short form 36 (SF-36). J Epidemiol Comm Health 49: 642–644
Julious SA, George SL, Machin D and Stephens RJ (1997) Sample sizes for randomised trials measuring quality of life in cancer patients. Quality of Life Research 6: 109–117
Machin D, Campbell MJ, Fayers PM and Pinol APY (1997) Statistical Tables for the Design of Clinical Studies. Blackwell Scientific: Oxford
Medical Research Council Lung Cancer Working Party (1996) Randomised trial of four-drug vs less intensive two-drug chemotherapy in the palliative treatment of patients with small-cell lung cancer (SCLC) and poor prognosis. Br J Cancer 73: 406–413
Moorey S, Greer S, Watson M, Gorman C, Rowden L, Tunmore R, Robertson B and Bliss J (1991) The factor structure and factor stability of the Hospital Anxiety and Depression Scale in patients with cancer. Br J Psych 158: 255–259
Whitehead J (1993) Sample size calculations for ordered categorical data. Stats in Med 12: 2257–2273
Zigmond AS and Snaith RP (1983) The Hospital Anxiety and Depression Scale. Acta Psychiatric Scand 67: 361–370
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Julious, S., Campbell, M., Walker, S. et al. Sample sizes for cancer trials where Health Related Quality of Life is the primary outcome. Br J Cancer 83, 959–963 (2000). https://doi.org/10.1054/bjoc.2000.1383
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1383
Keywords
This article is cited by
-
Power(ful) myths: misconceptions regarding sample size in quality of life research
Quality of Life Research (2022)