Abstract
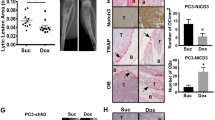
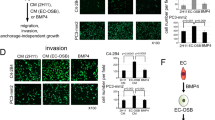
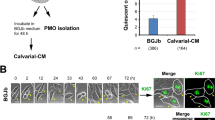
The causes for the propensity of metastasized prostate cancer cells to grow in bone and to induce osteoblastic lesions remain unresolved. Co-culture of human prostate cancer cell lines with bone slices was determined to increase the level of endothelin-1 (ET-1) mRNA and its production. ET-1 is an ejaculate protein that also stimulates osteoblasts. Osteoclastic bone resorption was significantly blocked by the presence of androgen-independent prostate cancer cells in a dose-dependent manner as that of synthetic ET-1. The inhibition could be neutralized by specific ET-1 antibody, indicating the association of prostate cancer-derived ET-1 with inhibition of bone resorption. The combined ET-1 activity on osteoclasts and osteoblasts disrupts bone remodelling. ET-1 production is also elevated in the presence of prostate-specific antigen (PSA). ET-1 in turn enhances DNA synthesis of prostate cancer cells. Interactions among cancer cells, bone, ET-1 and PSA may be critical in cancer growth and lesions in bone. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Abolhassani M and Chiao JW (1995) Antiproliferative effect of a prostate cell-derived activity on the human androgen-dependent prostatic carcinoma cell line LNCaP. J Interferon Cytokine Res 15: 179–185
Bagnato A (1999) Expression of endothelin 1 and endothelin A receptor in ovarian carcinoma: evidence for an autocrine role in tumour growth. Cancer Res 59: 720–727
Boyde A, Ali NH and Jones SI (1984) Resorption of dentine by isolated osteoclasts in vitro. Br Dent J 156: 216–220
Casey ML, Byrd W and MacDonald P (1992) Massive amounts of immunoreactive endothelin in human seminal fluid. J Clin Endocrinol Metab 74: 223–228
Chambers TJ (1984) Resorption of bone by isolated rabbit osteoclasts. J Cell Sci 66: 383–399
Charbon SA (1983) Histomorphometric analysis of sclerotic bone metastases from prostatic carcinoma with special reference to osteomalacia. Cancer 51: 918–927
Dempster DW, Marrills RJ, Herbert W and Arnett TR (1987) Biological activity of chicken calcitonin: effects on neonatal rat and embryonic chick osteoclasts. J Bone Mineral Res 2: 443–448
Horoszewicz JS (1983) LNCaP model of human prostatic carcinoma. Cancer Res 43: 1809–1818
Koutsilieris M (1987) Characteristics of prostate-derived growth factors for cells of the asteoblast phenotype. J Chem Invest 80: 941–948
Martinez J, Silva S and Santibanez JF (1996) Prostate-derived soluble factors block osteoblast differentiation in culture. J Cell Biochem 61: 18–25
Moonga BS and Chiao JW (1998) Detection of an inhibiting activity for osteoclastic bone resorption from human prostatic cancer cells. Cancer Lett 123: 15–20
Muraki J (1990) Establishment of a new human prostatic cancer cell line (JCA-1). Urology 36: 79–84
Nelson JB (1999) New bone formation in an osteoblastic tumour model is increased by endothelin 1 over expression and decreased by endothelin A receptor. Blockade Urology 53: 1063–1069
Nelson JB (1995) Identification of endothelin-1 in the pathophysiology of metastatic adenocarcinoma of the prostate. Nat Med 1: 944–949
Rabbani SA (1990) An amino-terminal fragment of urokinase isolated from a prostate cancer cell line (PC-3) is mitogenic for osteoblast-like cells. Biochem Biophys Res Commun 173: 1058–1064
Raff T (1997) Design and testing of beta-actin primers for RT-PCR that do not co amplify processed pseudogenes. Biotechnique 23: 456–460
Santibanez JF, Silva S and Martinez J (1996) Soluble factors produced by PC-3 prostate cells decrease collagen content and mineralization rate in fetal rat osteoblasts in culture. Br J Cancer 74: 418–422
Sharp WS and McDonald JR (1942) Reaction of bone to metastasis from carcinoma of the breast and prostate. Arch Pathol 17: 312–317
Shioide M and Noda M (1993) Endothelin modulates osteopontin and osteocalcin messenger RNA expression in rat osteoblastic osteosarcoma cells. J Cell Biochem 53: 176–180
Towhidul ASM (1992) Endothelin inhibits osteoclastic bone resorption by a direct effect on cell motility: implications for the vascular control of bone resorption. Endocrinology 130: 3617–3624
Wang LG, Liu XM, Kreis W and Budman DR (1996) Involvement of prostate specific antigen in the stimulation of LNCaP cell growth. Oncology Report 3: 911–917
Yanagisawa M (1994) The endothelin system. A new target for therapeutic intervention. Circulation 89: 1320–1322
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Chiao, J., Moonga, B., Yang, Y. et al. Endothelin-1 from prostate cancer cells is enhanced by bone contact which blocks osteoclastic bone resorption. Br J Cancer 83, 360–365 (2000). https://doi.org/10.1054/bjoc.2000.1261
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1261
Keywords
This article is cited by
-
Cancer-targeted therapies and radiopharmaceuticals
BoneKEy Reports (2015)
-
A phase I study of zibotentan (ZD4054) in patients with metastatic, castrate-resistant prostate cancer
Investigational New Drugs (2011)
-
The PCa Tumor Microenvironment
Cancer Microenvironment (2011)
-
Tumor-Stromal Interactions in Bone Metastasis
Current Osteoporosis Reports (2010)