Abstract
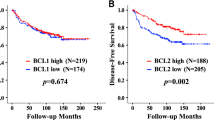
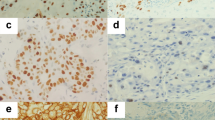
Photodynamic therapy (PDT) in early squamous cell carcinoma of the bronchus has been shown to result in complete response (CR) and cure. However, local recurrence after PDT develops frequently even after complete remission. Because the effect of PDT had been reported to depend on apoptosis, and apoptosis is inhibited by bcl-2 protein, the relationship between the expression of bcl-2 protein and local recurrence after PDT was examined immunohistochemically. From 1983 to 1997, 50 patients with 59 early squamous cell carcinoma of the bronchus received PDT, and a CR was obtained in 43 lesions (72.8%). As there was no recurrence among tumours that were disease-free for more than 2 years, in this study the tumours were defined as cured when recurrence did not occur 2 years subsequent to the receiving of PDT. Of these CR lesions, 31 carcinomas (53.4%) resulted in a cure. Bcl-2 immunoreactivity was detected in 23 tumours (46.9%) and p53 immunoreactivity was detected in 22 tumours (44.9%). When all tumours were divided into either a large tumour with a longitudinal tumour length of 10 mm or more, or a small tumour with a length of less than 10 mm, the large tumour expressed more bcl-2 protein than the small tumour (P = 0.0155). The degree of bcl-2 expression was significantly related with tumour size (P = 0.0155). The expression of bcl-2 and p53 protein was not associated with the cure rate due to PDT. Tumour length and T status in TNM staging were significantly related to the cure by univariate analysis. T status was the only predictor of the cure according to mutivariate analysis. Of 42 CR lesions, the expression of neither bcl-2 nor p53 protein was associated with local recurrence; only T status was significantly associated (P = 0.008). The relationship between the expression of oncoprotein and local recurrence after PDT was not documented in this study. The success of PDT may depend on the exact assessment of tumour size under optimized PDT illumination. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Agarwal ML, Clay ME, Harvey EJ, Evans HH, Antunez AR and Oleinick NL (1991) Photodynamic therapy induces rapid cell death by apoptosis in L5178Y mouse lymphoma cells. Cancer Res 51: 5993–5996
Agarwal ML, Larkin HE, Zaidi SIA, Mukhtar H and Oleinick NL (1993) Phospholipase activation triggers apoptosis in photosensitized mouse lymphoma cells. Cancer Res 53: 5897–5902
Bennet WP, Colby TV, Travis WD, Borkowski A, Jones RT, Lane DP, Metcalf RA, Samet JM, Takeshima Y, Gu JR, Vahakangas KH, Soini Y, Paakko P, Welsh JA, Trump BF and Harris CC (1993) P53 protein accumulates frequently in early bronchial neoplasia. Cancer Res 53: 4817–4822
Braichotte D, Savary JF, Glanzmann T, Monnier P, Wagnieres G and Bergh H (1996) Optimizing light dosimetry in photodynamic therapy of the bronchi by fluorescence spectroscopy. Lasers Med Sci 11: 247–254
Cortese DA, Edell ES and Kinsey JH (1997) Photodynamic therapy for early stage squamous cell carcinoma of the lung. Mayo Clin Proc 72: 595–602
Edell ES and Cortese DA (1992) Photodynamic therapy in the management of early superficial squamous cell carcinoma as an alternative to surgical resection. Chest 102: 1319–1322
Fisher AMR, Danenberg K, Banerjee D, Bertino JR, Danenberg P and Gomer CJ (1997) Increased photosensitivity in HL60 cells expressing wild-type p53. Photochem. Photobiol 66: 265–270
Furuse K, Fukuoka M, Kato H, Horai T, Kubota K, Kodama N, Kusunoki Y, Takifuji N, Okunaka T, Konaka C, Wada H and Hayata Y (1993) A prospective phase II study on photodynamic therapy with photofrin II for centrally located early-stage lung cancer. J Clin Oncol 11: 1852–1857
He J, Agarwal ML, Larkin HE, Friedman LR, Xue L and Oleinick NL (1996) The induction of partial resistance to photodynamic therapy by the protooncogene bcl-2. Photochem Photobiol 64: 845–852
He X, Sikes RA, Thomsen S, Chung LWK and Jacques SL (1994) Photodynamic therapy with photofrin II induces programmed cell death in carcinoma cell lines. Photochem Photobiol 59: 468–473
Kato H, Konaka C, Kawate N, Shinohara H, Kinoshita K, Noguchi M, Ootomo S and Hayata Y (1986) Five-year disease-free survival of a lung cancer patient treated only photodynamic therapy. Chest 90: 768–770
Kawaguchi T, Furuse K, Kawahara M, Yamamoto S and Sutedja TG (1998) Histological examination of bronchial mucosa after photodynamic therapy showing no selectivity of effect between tumor and normal mucosa. Laser Med Sci 13: 265–270
Koukourakis MI, Giatromanolaki A, O’Byrne KJ, Whitehouse RM, Talbot DC, Gatter KC and Harris AL (1997) Potential role of bcl-2 as a suppressor of tumour angiogenesis in non-small-cell lung cancer. Int J Cancer 74: 565–570
Luo Y and Kessel D (1997) Initiation of apoptosis versus necrosis by photodynamic therapy with chloroaluminium phthalocyanine. Photochem Photobiol 66: 479–483
Mountain CF (1986) A new international staging system for lung cancer. Chest 89: 225s–233s
Nishio M, Koshikawa T, Kuroishi T, Suyama M and Uchida K (1996) Prognostic significance of abnormal p53 accumulation in primary, resected non-small-cell lung cancers. J Clin Oncol 14: 497–502
Pass HI (1993) Photodynamic therapy in oncology: mechanisms and clinical use. J Natl Cancer Inst 85: 443–456
Salgia R and Skarin AT (1998) Molecular abnormalities in lung cancer. J Clin Oncol 16: 1207–1217
Separovic D, He J and Oleinick NL (1997) Ceramide generation in response to photodynamic treatment of L5178Y mouse lymphoma cells. Cancer Res 57: 1717–1721
Star WM, Marijnissen H, van den Berg Blok E, Versteeg JAC, Franken KA and Reinhold HS (1986) Destruction of rat mammary tumor and normal tissue microcirculation by hematoporphyrin derivative photoradiation observed in vivo in sandwich observation chamber. Cancer Res 46: 2532–2540
Sutedja G, Lam S, LeRiche JC and Postmus PE (1994) Response and pattern of failure after photodynamic therapy for intraluminal stage I lung cancer. J Bronchol 1: 295–298
Sutedja G and Postmus PE (1996) Photodynamic therapy in lung cancer. A review. Photochem Photobiol 36: 199–204
Usuda K, Saito Y, Nagamoto N, Sato M, Sagawa M, Kanma K, Takahashi S, Endo C and Fujimura S (1993) Relation between bronchoscopic findings and tumor size of roentgenographically occult bronchogenic squamous cell carcinoma. J Thorac Cardiovasc Surg 106: 1098–1103
Walker C, Robertson L, Myskow M and Dixon G (1995) Expression of the bcl-2 protein in normal and dysplastic bronchial epithelium and in lung carcinomas. Br J Cancer 72: 164–169
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Kawaguchi, T., Yamamoto, S., Naka, N. et al. Immunohistochemical analysis of Bcl-2 protein in early squamous cell carcinoma of the bronchus treated with photodynamic therapy. Br J Cancer 82, 418–423 (2000). https://doi.org/10.1054/bjoc.1999.0936
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.1999.0936
Keywords
This article is cited by
-
The study of effect and mechanism of 630-nm laser on human lung adenocarcinoma cell xenograft model in nude mice mediated by hematoporphyrin derivatives
Lasers in Medical Science (2020)
-
Differential cytotoxic response in keloid fibroblasts exposed to photodynamic therapy is dependent on photosensitiser precursor, fluence and location of fibroblasts within the lesion
Archives of Dermatological Research (2012)
-
In vitro and in vivo inhibition of epidermal growth factor receptor-tyrosine kinase pathway by photodynamic therapy
Oncogene (2001)