Summary
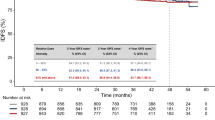
The purpose of this study was to examine the association between the leucocyte nadir and prognosis in breast cancer patients receiving adjuvant chemotherapy consisting of cyclophosphamide, methotrexate and fluorouracil (CMF). Three hundred and sixty-eight patients with node-positive breast cancer without distant metastases were treated with six cycles of adjuvant CMF. Some patients (n = 60) also received tamoxifen. All patients underwent surgery and received radiotherapy to the axillary and supraclavicular lymph nodes and the chest wall. The effect of leucopenia caused by CMF on distant disease-free survival (DDFS) and overall survival (OS) was assessed. A low leucocyte nadir during the chemotherapy was associated with a long DDFS in univariate analysis when tested as a continuous variable (the relative risk (RR) 1.3, 95% confidence interval (CI) 1.04–1.06, P = 0.02). Similarly, when the leucocyte nadir count was divided into tertiles, the patients who had the highest nadir values during the six-cycle treatment had worst outcome (RR 1.6, 95% CI 1.07–2.5, P = 0.02). However, in a multivariate analysis only the number of affected lymph nodes, tumour size, progesterone receptor status, surgical procedure, age and adjuvant tamoxifen therapy retained prognostic significance, whereas the leucocyte nadir count did not. A low leucocyte nadir during the adjuvant CMF chemotherapy is associated with favourable DDFS and it may be a useful biological marker for chemotherapy efficacy.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Ahmann, D. L., O’Fallon, J. R., Scanlon, P. W., Payne, W. S., Bisel, H. F., Edmonson, J. H., Frytak, S., Hahn, R. G., Ingle, J. N., Rubin, J. & Creagan, E. T. (1982). A preliminary assessment of factors associated with recurrent disease in a surgical adjuvant clinical trial for patients with breast cancer with special emphasis on the aggressiveness of therapy. Am J Clin Oncol 5: 371–381.
Ang, P. T., Buzdar, A. U., Smith, T. L., Kau, S. & Hortobagyi, G. N. (1989). Analysis of dose intensity in doxorubicin-containing adjuvant chemotherapy in stage II and III breast carcinoma. J Clin Oncol 7: 1677–1684.
Bonadonna, G. & Valagussa, P. (1981). Dose–response effect of adjuvant chemotherapy in breast cancer. N Engl J Med 304: 10–15.
Colleoni, M., Price, K., Castiglione-Gertch, A., Goldhirch, A. & Coates, A. (1998). Dose–response effect of adjuvant cyclophosphamide, methotrexate, 5-fluorouracil (CMF) in node-positive breast cancer. Eur J Cancer 34: 1693–1700.
Cox, D. (1972). Regression models and life tables. J R Statist Soc Ser B34: 187–220.
Early Breast Cancer Trialists’ Collaborative Group (1992). Systemic treatment of early breast cancer by hormonal, cytotoxic, or immune therapy: 133 randomised trials involving 31 000 recurrences and 24 000 deaths among (1999) 75 000 women. Lancet 339: 1–15
Fisher, B., Anderson, S., Wickerham, D. L., DeCillis, A., Dimitrov, N., Mamounas, E., Wolmark, N., Pugh, R., Atkins, J. N., Meyers, F. J., Abramson, N., Wolter, J., Bornstein, R. S., Levy, L., Romond, E. H., Caggiano, V., Grimaldi, M., Jochimsen, P. & Deckers, P. (1997). Increased intensification and total dose of cyclophosphamide in a doxorubicin-cyclophosphamide regimen for the treatment of primary breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-22. J Clin Oncol 15: 1858–1869.
Fumoleau, P., Devaux, Y., Vo, V. M., Kerbrat, P., Fargeot, P., Schraub, S., Mihura, J., Namer, M. & Mercier, M. (1993). Premenopausal patients with node-positive resectable breast cancer. Preliminary results of a randomised trial comparing 3 adjuvant regimens: FEC 50 × 6 cycles vs FEC 50 × 3 cycles vs FEC 75 × 3 cycles. The French Adjuvant Study Group. Drugs 2: 38–45.
Glucksberg, H., Rivkin, S. E., Rasmussen, S., Tranum, B., Gad, E. l., Mawla, N., Costanzi, J., Hoogstraten, B., Athens, J., Maloney, T., McCracken, J. & Vaughn, C. (1982). Combination chemotherapy (CMFVP) versus L-phenylalanine mustard (L-PAM) for operable breast cancer with positive axillary nodes: a Southwest Oncology Group Study. Cancer 50: 423–434.
Howell, A., Rubens, R. & Bush, H. (1984). A controlled trial of adjuvant chemotherapy with melphalan versus cyclophosphamide, methotrexate and fluorouracil for breast cancer. Resent Results Cancer Res 96: 74–89.
Kaplan, E. & Meier, P. (1958). Non-parametric estimation from incomplete observations. J Am Statist Assoc 53: 457–481.
Longo, D. L., Duffey, P. L., DeVita, V. J., Wesley, M. N., Hubbard, S. M. & Young, R. C. (1991). The calculation of actual or received dose intensity: a comparison of published methods. J Clin Oncol 9: 2042–2051.
Mouridsen, H. T., Rose, C., Brincker, H., Thorpe, S. M., Rank, F., Fischerman, K. & Andersen, K. W. (1984). Adjuvant systemic therapy in high-risk breast cancer: the Danish Breast Cancer Cooperative Group’s trials of cyclophosphamide or CMF in premenopausal and tamoxifen in postmenopausal patients. Recent Results Cancer Res 96: 117–128.
Peto, R., Pike, M. & Armitage, P. (1977). Dosing and analysis of randomized clinical trials requiring prolonged observation of each patient. II. Analysis and examples. Br J Cancer 35: 1–39.
Pronzato, P., Campora, E., Amoroso, D., Bertelli, G., Botto, F., Conte, P. F., Sertoli, M. R. & Rosso, R. (1989). Impact of administration-related factors on outcome of adjuvant chemotherapy for primary breast cancer. Am J Clin Oncol 12: 481–485.
Redmond, C., Fisher, B. & Wieand, H. S. (1983). The methodologic dilemma in retrospectively correlating the amount of chemotherapy received in adjuvant therapy protocols with disease-free survival. Cancer Treatment Rep 67: 519–526.
Rodriguez, K. R., Hortobagyi, G. N., Buzdar, A. U. & Blumenschein, G. R. (1981). Combination chemotherapy for breast cancer metastatic to bone marrow. Cancer 48: 227–232.
Saarto, T., Blomqvist, C., Rissanen, P., Auvinen, A. & Elomaa, I. (1997). Haematological toxicity: a marker of adjuvant chemotherapy efficacy in stage II and III breast cancer. Br J Cancer 75: 301–305.
Senn, H., Jungi, W. & Amgewerd, R. (1984). Adjuvant chemoimmunotherapy with LMF + BCG in node-negative and node positive breast cancer. 8 year results. Recent Results Cancer Res 96: 90–101.
Tormey, D., Gelman, R. & Falkson, G. (1983). Prospective evaluation of rotating chemotherapy in advanced breast cancer. An Eastern Cooperative Oncology Group Trial. Am J Clin Oncol 6: 1–18.
Velez-Garcia, E., Carpenter, J. & Moore, M. (1987). Postsurgical adjuvant chemotherapy with or without radiotherapy in women with breast cancer and positive axillary nodes: Progress report of Southeastern Cancer Study Group (SEG) trial. Adjuvant Ther Cancer V: 347–355.
Wood, W. C., Budman, D. R., Korzun, A. H., Cooper, M. R., Younger, J., Hart, R. D., Moore, A., Ellerton, J. A., Norton, L., Ferree, C. R., Colangelo Ballow, A., Frei, III E. & Henderson, C. (1994). Dose and dose intensity of adjuvant chemotherapy for stage II, node-positive breast carcinoma. N Engl J Med 330: 1253–1259.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Poikonen, P., Saarto, T., Lundin, J. et al. Leucocyte nadir as a marker for chemotherapy efficacy in node-positive breast cancer treated with adjuvant CMF. Br J Cancer 80, 1763–1766 (1999). https://doi.org/10.1038/sj.bjc.6690594
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690594
Keywords
This article is cited by
-
Optimization of clinical dosing schedule to manage neutropenia: learnings from semi-mechanistic modeling simulation approach
Journal of Pharmacokinetics and Pharmacodynamics (2020)
-
Long-term safety and survival outcomes from the Scandinavian Breast Group 2004-1 randomized phase II trial of tailored dose-dense adjuvant chemotherapy for early breast cancer
Breast Cancer Research and Treatment (2018)
-
Leucopenia and treatment efficacy in advanced nasopharyngeal carcinoma
BMC Cancer (2015)
-
Neutrophil-guided dosing of anthracycline–cyclophosphamide-containing chemotherapy in patients with breast cancer: a feasibility study
Medical Oncology (2015)
-
Randomised phase II study comparing dose-escalated weekly paclitaxel vs standard-dose weekly paclitaxel for patients with previously treated advanced gastric cancer
British Journal of Cancer (2014)