Abstract
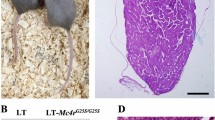
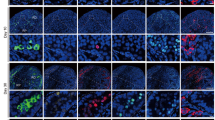
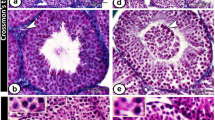
An animal model of experimental testicular teratoma has been established to study how a teratoma affects the host testis and how the host testis reacts against the teratoma. 129/SvJ-mice were used as experimental animals. To induce the experimental testicular teratoma, male gonadal ridges from 12-day-old 129/SvJ-mouse fetuses were grafted into the testes of adult mice for 1–12 weeks. The developing tumour was analysed by light and electron microscopy and by immunocytochemical localization of transcription factors SOX9 and c-kit, glial fibrillary acidic protein (GFAP) and type IV collagen. Testicular teratoma was observed in 36 out of 124 testes with implanted fetal gonadal ridges (frequency 29%). One spontaneous testicular teratoma was observed in this material from 70 male mice (1.5%). One week after implantation intracordal clusters of cells were seen in embryonic testicular cords of the graft as the first sign of testicular teratomas. Four weeks after implantation the embryonic testicular cords had totally disappeared from grafts with teratomas, and the tumour tissue had enlarged the testis and invaded the interstitium of the host testis. It consisted of solitary pieces of immature cartilage as well as of glial cells and of primitive neuroepithelium. Six to eight weeks after implantation the tumour tissue had expanded so that the enlarged testis could be detected by macroscopic enlargement of the scrotum. The testicular tissue of the host had practically disappeared, and only solitary disrupted seminiferous tubules of the host were seen surrounding the teratoma. Neuroepithelial structures of some teratomas cultured for 8 weeks had cells with a granular nucleus as a sign of obvious apoptosis. Eleven to 12 weeks after implantation the growth of the teratoma had stopped, and the histology corresponded to that of a mature cystic teratoma. GFAP, SOX9 and type IV collagen were strongly positive in some parts of the tumours cultured for 4 and 8 weeks, while only occasional c-kit-positive areas were observed in tumours cultured for 8 weeks. As conclusions: (1) the metastasizing capacity of the experimental testicular teratoma is very low during 12 weeks, but the behaviour of the tumour in the testicular tissue of the graft is invasive; (2) the growth of experimental testicular teratomas cease 6–8 weeks after implantation of the fetal gonadal ridges with the obvious apoptosis of the immature tissue components; (3) the model of experimental testicular teratoma in the mouse is suitable for studying how the teratoma affects the host testis and how the host testis reacts to teratoma.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Adami, H. O., Bergström, R., Möhner, M., Zatonski, W., Storm, H., Ekbom, A., Tretli, S., Teppo, L., Ziegler, H., Rahu, M., Gurevicius, R. & Stengrevics, A. (1994) Testicular cancer in nine Northern European countries. Int J Cancer 59: 33–38.
Andrews, P. W. (1998) Teratocarcinomas and human embryology: pluripotent human EC cell lines. APMIS 106: 158–168.
Andrews, P. W., Casper, J., Damjanov, I., Duggan-Keen, M., Giwercman, A., Hata, J. I., von Keitz, A., Looijenga, L. H. J., Millan, L. J., Oosterhuis, J. W., Pera, M., Sawada, M., Schmoll, H. J., Skakkebaek, N. E., van Putten, W. & Stern, P. (1996) Comparative analysis of cell surface antigens expressed by cell lines derived from human germ cell tumours. Int J Cancer 66: 806–816.
Carlsen, E., Giwercman, A., Keiding, N. & Skakkebaek, N. E. (1992) Evidence for decreasing quality of semen during past 50 years. Br Med J 305: 609–613.
Finch, B. W. & Ephrussi, B. (1967) Retention of multiple developmental potetialites by cells of a mouse testicular teratocarcinoma during prolonged culture in vitro and their extinction upon hybridization with cells of permanent lines. Proc Natl Acad Sci USA 57: 615–621.
Foidart, J. M., Berman, J. J., Paglia, L., Rennard, S., Abe S Perantoni, A. & Martin, G. R. (1980) Synthesis of fibronectin, laminin, and several collagens by a liver-derived epithelial line. Lab Invest 42: 525–532.
Foster, J. W., Dominguez-Steglich, M. A., Guioli, s., Kowk, G., Weller, P. A., Stevanovic, M., Weissenbach, J., Mansour, S., Young, I. D., Goodfellow, P. N., Brook, J. D. & Schafer, A. J. (1994) Campomelic dysplasia and autosomal sex reversal caused by mutations in an Sry-related gene. Nature 372: 525–530.
Friedrich, T. D., Regenass, U. & Stevens, L. C. (1983) Mouse genital ridges in organ culture: the effects of temperature on maturation and experimental induction of teratocarcinogenesis. Differentiation 24: 60–64.
Fröjdman, K., Pelliniemi, L. J. & Virtanen, I. (1998) Differential distribution of type IV collagen chains in the developing rat testis and ovary. Differentiation 63: 125–130.
Gilliland, F. D. & Key, C. R. (1995) Male genital cancers. Cancer 75: 295–315.
Hsu, S. M., Raine, L. & Fanger, H. (1981) A comparative study of the peroxidase–antiperoxidase method and an avidin-biotin complex method for studying polypeptide hormones with radioimmunoassay antibodies. Am J Clin Pathol 75: 734–738.
Karnovsky, M. J. (1971) Use of ferrocyanide-reduced osmium tetroxide in electron microscopy. J Cell Biol 284: 146
Klepp, O., Olsson, A. M., Henrikson, O., Aass, N., Dahl, O., Stenwig, A. E., Persson, B. E., Cavallin-Stahl, E., Fossa, S. D. & Wahlqvist, L. (1990) Prognostic factors in clinical stage I non-seminomatous germ cell tumors of the testis: multivariate analysis of a prospective multicenter study. Swedish–Norwegian Testicular Cancer Group. J Clin Oncol 8: 509–518.
Martin, G. R. & Evans, M. J. (1974) The morphology and growth of a pluripotent teratocarcinoma cell line and its derivatives in tissue culture. Cell 2: 163–172.
Matin, A., Collin, G. B., Varnum, D. S. & Nadeau, J. H. (1998) Testicular teratocarcinogenesis in mice – a review. APMIS 106: 174–182.
McLean, I. W. & Nakane, P. K. (1974) Periodate-lysine-paraphormaldehyde fixative. A new fixative for immunoelectron microscopy. J Histochem 22: 1077–1083.
Morais da Silva, S., Hacker, A., Harley, V., Goodfellow, P., Swain, A. & Lovell-Badge, R. (1996) SOX9 expression during gonadal development implies a conserved role for the gene in testis differentiation in mammals and birds. Nature Genet 14: 62–68.
Mostofi, F. K. & Price, E. B. (1973). Tumors of the testis. In Tumors of the Male Genital System. Atlas of Tumor Pathology, 2nd series, Fasc. 8, p. 2. Armed Forces Institute of Pathology: Washington DC
Mäenpää, A., Kovanen, P. E., Paetau, A., Jääskeläinen, J. & Timonen, T. (1997) Lymphocyte adhesion molecule ligands and extracellular matrix proteins in gliomas and normal brain: expression of VCAM-1 in gliomas. Acta Neuropathol 94: 216–225.
Noguchi, T. & Stevens, L. C. (1982) Primordial germ cell proliferation in fetal testis in mouse strains with high and low incidences of congenital testicular teratomas. J Natl Cancer Inst 69: 907–913.
Oosterhuis, J. W., Castedo, SMMJ, De Jong, B., Cornelisse, C. J., Dam, A., Sleijfer, D. T. & Schrafford Koops, H. (1989) Ploidy of primary germ cell tumours of the testis. Lab Invest 60: 14–21.
Rajpert-De Meyts, E. & Skakkebaek, N. E. (1994) Expression of c-kit protein product in carcinoma-in-situ and invasive testicular germ cell tumours. Int J Androl 17: 85–92.
Ro, J. Y., Dexeus, F. H., el-Naggar, A. & Ayala, A. G. (1991) Testicular germ cell tumors. Clinically relevant pathologic findings. Pathol Annu 26: 59–87.
Solter, D. & Damjanov, I. (1979) Teratocarcinoma and the expression of oncodevelopmental genes. Methods Cancer Res 18: 277–232.
Solter, D., Skreb, N. & Damjanov, I. (1970) Extra-uterine growth of mouse egg-cylinders results in malignant teratoma. Nature 227: 503–504.
Stempak, J. G. & Ward, R. T. (1964) An improved staining method for electron microscopy. J Cell Biol 22: 697–701.
Stevens, L. C. (1958) Studies on transplantable testicular teratomas of strain 129 mice. J Natl Cancer Inst 20: 1257–1275.
Stevens, L. C. (1964) Experimental production of testicular teratomas in mice. Proc Natl Acad Sci USA 52: 654–661.
Stevens, L. C. (1967a). The biology of teratomas. In Advances in Morphogenesis, Abercrombie M and Brachet J (eds., p. 1–131). Academic Press: New York
Stevens, L. C. (1967b) Origin of testicular teratomas from primordial germ cells in mice. J Natl Cancer Inst 38: 549–552.
Stevens, L. C. (1970a) Experimental production of testicular teratomas in mice of strains 129, A/He, and their F1 hybrids. J Natl Cancer Inst 44: 923–929.
Stevens, L. C. (1970b) The development of transplantable teratocarcinomas from intratesticular grafts of pre-and postimplantation mouse embryos. Dev Biol 21: 364–382.
Stevens, L. C. (1973) A new inbred subline of mice (129/terSv) with a high incidence of spontaneous congenital testicular teratomas. J Natl Cancer Inst 50: 235–239.
Stevens, L. C. (1984) Spontaneous and experimentally induced testicular teratomas in mice. Cell Differ 15: 69–74.
Stevens, L. C. & Mackensen, J. A. (1961) Genetic and environmental influences on testicular teratocarcinogenesis in mice. J Natl Cancer Inst 27: 443–453.
Sundström, J., Veräjänkorva, E., Salminen, E., Pelliniemi, L. J. & Pöllänen, P. (1998). Experimental testicular teratoma promotes formation of humoral immune responses in the host testis. J Reprod Immunol (in press)
Towbin, H., Staehelin, T. & Gordon, J. (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76: 4350–4354.
Van Gurp, RJLM, Oosterhuis, J. W., Kalscheuer, V., Mariman, E. C. M. & Looijenga, L. H. J. (1994) Human testicular germ cell tumours show biallelic expression of the H19 and IGF2 gene. J Natl Cancer Inst 86: 1070–1077.
Venable, J. H. & Coggeshall, R. A. (1965) Simplified lead citrate stain for use in electron microscopy. J Cell Biol 25: 407–408.
Walt, H., Oosterhuis, J. W. & Stevens, L. C. (1993) Experimental testicular germ cell tumorogenesis in mouse strains with and without spontaneous tumours differs from development of germ cell tumours of the adult human testis. Int J Androl 16: 267–271.
Wang, D. & Enders, G. C. (1996) Expression of a specific mouse germ cell nuclear antigen (GCNA1) by early embryonic testicular teratoma cells in 129/Sv-Sl/+ mice. Cancer Lett 100: 31–36.
Zhao, Q., Eberspaecher, H., Lefebve, V. & De Crombrugghe, B. (1997) Parallel expression of SOX9 and Col2a1 in cells undergoing chondrogenesis. Dev Dyn 209: 377–386.
Zheng, T., Holford, T. R., Ma, Z., Ward, B. A., Flannery, J. & Boyle, P. (1996) Continuing increase in incidence of germ-cell testis cancer in young adults: experience from Connecticut, USA, 1935–1992. Int J Cancer 65: 723–729.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Sundström, J., Pelliniemi, L., Kuopio, T. et al. Characterization of the model for experimental testicular teratoma in 129/SvJ-mice. Br J Cancer 80, 149–160 (1999). https://doi.org/10.1038/sj.bjc.6690334
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690334