Abstract
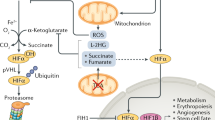
Cellular responses to hypoxia include modulation of respiration rate and up-regulation of genes which encode for angiogenesis factors. We tested whether human malignant glioma cells vary in their response to hypoxic stress over the range of oxygen concentrations which exist in tumours. In five cell lines tested, decreased oxygen availability resulted in decreased rates of oxygen utilization, however substantial differences in the magnitude of the response were observed. Northern blot analysis was used to study induction of vascular endothelial growth factor mRNA in response to hypoxia. In two cell lines, modest hypoxia increased vascular endothelial growth factor mRNA levels compared with those of aerobic controls. In two additional cell lines, vascular endothelial growth factor mRNA was constituitively expressed under aerobic conditions and was not further increased by hypoxia. These findings demonstrate that differences in the response to hypoxia exist among human malignant glioma cell lines and suggest that therapies designed to exploit tumour hypoxia may have varying effects in tumours with different hypoxic stress responses.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Allalunis-Turner, M. J. & Siemann, D. W. (1986) Characterization of human tumor xenografts using different enzyme dissociation techniques. Br J Cancer 54: 615–622.
Allalunis-Turner, M. J., Barron, G. M., Day, R. S., III, Fulton, D. S. & Urtasun, R. C. (1992) Radiosensitivity testing of human primary brain tumor specimens. Int J Radiat Oncol Biol Phys 23: 339–343.
Berkman, R. A., Merrill, M. J., Reinhold, W. C., Monacci, W. T., Saxena, A., Clark, W. C., Robertson, J. T., Ali, I. U. & Oldfield, E. H. (1993) Expression of the vascular permeability factor/vascular endothelial growth factor gene in central nervous system neoplasms. J Clin Invest 91: 153–159.
Berse, B., Brown, L. F., Van De Water, L., Dvorak, H. F. & Senger, D. R. (1992) Vascular permeability factor (vascular endothelial growth factor) gene is expressed differentially in normal tissues, macrophages and tumors. Mol Biol Cell 3: 211–220.
Boag, J. W. (1970) Cell respiration as a function of oxygen tension. Int J Radiat Biol 5: 475–478.
Brooks, D. (1990). In-vivo metabolism of human cerebral tumors. In Neuro-Oncology: Primary Malignant Brain Tumors, Thomas D (ed.), pp. 122–134. Johns Hopkins University Press: Baltimore
Bussolino, F., Mantovani, A. & Persico, G. (1997) Molecular mechanisms of blood vessel formation. Trends Biol Sci 22: 251–256.
Chandel, N. S., Budinger, G. R. S., Choe, S. H. & Schumaker, P. T. (1997) Cellular respiration during hypoxia. J Biol Chem 272: 18808–18816.
Dewhirst, M. W., Secomb, T. W., Ong, E. T., Hsu, R. & Gross, J. F. (1994) Determination of local oxygen consumption rates in tumors. Cancer Res 54: 3333–3336.
Dvorak, H. F., Brown, L. F., Detmar, M. & Dvorak, A. M. (1995) Vascular permeability factor/vascular endothelial growth factor, microvascular hyperpermeability, and angiogenesis. Am J Pathol 146: 1029–1039.
Ema, M., Taya, S., Yokotani, N., Sogawa, K., Matsuda, Y. & Fujii-Kuriyama, Y. (1997) A novel bHLH-PAS factor with close sequence similarity to hypoxia-inducible factor 1a regulates the VEGF expression and is potentially involved in lung and vascular development. Proc Natl Acad Sci USA 94: 4273–4278.
Erecinska, M. & Wilson, D. F. (1982) Regulation of cellular energy metabolism. J Membr Biol 70: 1–14.
Ferrara, N., Houck, K., Jakeman, L. & Leung, D. W. (1992) Molecular and biological properties of the vascular endothelial growth factor family of proteins. Endocrinol Rev 13: 18–29.
Fontanini, G., Vignati, S., Boldrini, L., Chiné, S., Silvestri, V., Lucchi, M., Mussi, A., Angeletti, A. A. & Bevilacqua, G. (1997) Vascular endothelial growth factor is associated with neovascularization and influences progression of non-small cell lung carcinoma. Clin Cancer Res 3: 861–865.
Froese, G. (1962) The respiration of ascites tumor cells at low oxygen concentrations. Biochim Biophys Acta 57: 509–519.
Hanahan, D. & Folkman, J. (1996) Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 86: 353–364.
Hochachka, P. W. & Guppy, M. (1987). Animal anaerobes. In Metabolic Arrest and the Control of Biological Time, pp. 10–35. Harvard University Press: Cambridge
Hochachka, P. W., Buck, L. T. & Land, S. C. (1996) Unifying theory of hypoxia tolerance: molecular/metabolic defense and rescue mechanisms for surviving oxygen lack. Proc Natl Acad Sci USA 93: 9493–9498.
Houck, K. A., Leung, D. W., Rowland, A. M., Winter, J. & Ferrara, N. (1992) Dual regulation of vascular endothelial growth factor bioavailability by genetic and proteolytic mechanisms. Biol Chem 267: 26031–26037.
Iliopoulos, O., Levy, A. P., Jiang, C., Kaeling, W. G. Jr & Goldberg, M. A. (1996) Negative regulation of hypoxia-inducible genes by the von Hippel-Lindau protein. Proc Natl Acad Sci USA 93: 10595–10599.
Koch, C. J. (1984) A ‘thin film’ culturing technique allowing rapid gas-liquid equilibrium (6 seconds) with no toxicity to mammalian cells. Radiat Res 97: 434–438.
Koch, C. J., Howell, R. L. & Biaglow, J. E. (1979) Ascorbate anion potentiates the cytotoxicity of nitro-aromatic compounds under hypoxic and anoxic conditions. Br J Cancer 39: 321–329.
Lammertsma, M. A., Wise, R., Cox, T., Thomas, D. & Jones, T. (1985) Measurement of blood flow, oxygen utilization, oxygen extraction ratio and fractional blood volume in human brain tumours and surrounding oedematous tissue. Br J Radiol 58: 725–734.
Leith, J. T. & Michelson, S. (1995) Secretion rates and levels of vascular endothelial growth factor in clone A or HCT-8 human colon tumour cells as a function of oxygen concentration. Cell Prolif 28: 415–430.
Levy, A. P., Levy, N. S. & Goldberg, M. A. (1996) Post-transcriptional regulation of vascular endothelial growth factor by hypoxia. J Biol Chem 271: 2746–2653.
Levy, A. P., Levy, N. S., Iliopoulos, O., Jiang, C., Kaelin, W. G. Jr & Goldberg, M. A. (1997) Regulation of vascular endothelial growth factor by hypoxia and its modification by the von-Hippel-Lindau tumor suppressor gene. Kidney Int 51: 575–578.
Ohta, Y., Endo, Y., Tanaka, M., Shimizu, J., Oda, M., Hayashi, Y., Watanabe, Y. & Sasaki, T. (1996) Significance of vascular endothelial growth factor messenger RNA expression in primary lung cancer. Clin Cancer Res 2: 1411–1416.
Parliament, M. P., Franko, A. J., Allalunis-Turner, M. J., Mielke, B. W., Santos, C. L., Wolokoff, B. G. & Mercer, J. R. (1997) Anomalous patterns of nitroimidazole binding adjacent to necrosis in human glioma xenografts: possible role of decreased oxygen consumption. Br J Cancer 75: 311–318.
Plate, K. H., Breier, G., Weich, H. A. & Risau, W. (1992) Vascular endothelial growth factor is a potential tumour angiogenesis factor in human gliomas in vivo. Nature 359: 845–848.
Plate, K. H., Breier, G., Weich, H. A., Mennel, H. D. & Risau, W. (1994) Vascular endothelial growth factor and glioma angiogenesis: coordinate induction of VEGF receptors, distribution of VEGF protein and possible in vitro regulatory mechanisms. Int J Cancer 59: 520–529.
Salver, P., Mäenpää, H., Orpana, A., Alitalo, K. & Joensuu, H. (1997) Serum vascular endothelial growth factor is often elevated in disseminated cancer. Clin Cancer Res 3: 647–651.
Secomb, T. W., Hsu, R., Ong, E. T., Gross, J. F. & Dewhirst, M. W. (1995) Analysis of the effects of oxygen supply and demand on hypoxic fraction in tumors. Acta Oncol 34: 313–316.
Suzuki, K., Hayashi, N., Miyamoto, Y., Yamamoto, M., Ohkawa, K., Ito, Y., Sasaki, Y., Yamaguchi, Y., Nakase, H., Noda, K., Enomoto, N., Arai, K., Yamada, Y., Yoshihara, H., Tujimura, T., Kawano, K., Yoshikawa, K. & Kamada, T. (1996) Expression of vascular permeability factor/vascular endothelial growth factor in human hepatocellular carcinoma. Cancer Res 56: 3004–3009.
White, F. C., Carroll, S. M. & Kamps, M. P. (1995) VEGF mRNA is reversibly stabilized by hypoxia and persistently stabilized in VEGF-overexpressing human tumor cell lines. Growth Factors 12: 289–301.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Allalunis-Turner, M., Franko, A. & Parliament, M. Modulation of oxygen consumption rate and vascular endothelial growth factor mRNA expression in human malignant glioma cells by hypoxia. Br J Cancer 80, 104–109 (1999). https://doi.org/10.1038/sj.bjc.6690328
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690328
Keywords
This article is cited by
-
Why one-size-fits-all vaso-modulatory interventions fail to control glioma invasion: in silico insights
Scientific Reports (2016)
-
Hypoxic regulation of cytoglobin and neuroglobin expression in human normal and tumor tissues
Cancer Cell International (2010)
-
Graded hypoxia modulates the invasive potential of HT1080 fibrosarcoma and MDA MB231 carcinoma cells
Clinical & Experimental Metastasis (2008)
-
Variation in mitochondrial function in hypoxia-sensitive and hypoxia-tolerant human glioma cells
British Journal of Cancer (2002)