Summary
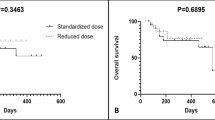
Skin deposits from breast cancer can present serious therapeutic problems, especially when resistant to conventional therapy. Topical application of a cytotoxic drug may represent an attractive new treatment modality devoid of major systemic toxicity. Miltefosine was selected because of its efficacy in breast cancer models. A mixture of alkylated glycerols of various chain lengths and water was used as the pharmaceutical vehicle to dissolve and to further facilitate tissue penetration of miltefosine. In our Institute a phase II study was performed to determine the efficacy and tolerability of topically applied miltefosine in patients with cutaneous metastases from breast cancer. Thirty-three patients in total entered the trial. A 6% miltefosine solution was applied once daily in the first week and twice daily in the following weeks. The planned minimum treatment duration was 8 weeks. We found an overall response rate of 43% for 30 evaluable patients, composed of 23% complete response and 20% partial response. The median response duration was 18 weeks, range 8–68. Toxicity consisted mainly of localized skin reactions, which could be controlled by a paraffin-based skin cream and, where appropriate, by dose modification. No systemic toxicities were observed. We conclude that topical miltefosine is an effective treatment modality in patients with skin metastases from breast cancer.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Ando, K., Kodama, K., Kato, A., Tamura, G. & Arima, K. (1972). Antitumor activity of glycerylethers. Cancer Res, 32: 125–129.
Berkovic, D., Grunwald, U., Menzel, W., Unger, C., Hiddeman, W. & Fleer, E. A. (1995). Effects of hexadecylphosphocholine on membrane phospholipid metabolism in human tumour cells. Eur J Cancer, 31A: 2080–2085.
Clive, S. & Leonard, R. C. F. (1997). Miltefosine in recurrent cutaneous breast cancer. Lancet, 349: 621–622.
Detmar, M., Geilen, C. C., Wieder, T., Orfanos, C. E. & Reuter, W. (1994). Phospholipid analogue hexadecylphosphocholine inhibits proliferation and phosphatidylcholine biosynthesis of human epidermal keratinocytes in vitro. J Inv Derm, 102: 490–494.
Dummer, R., Krasovec, M., Roeger, J., Sindermann, H. & Burg, G. (1993). Topical administration of hexadecylphosphocholine in patients with cutaneous lymphomas: results of a phase I/II study. J Am Academy Derm 29: 963–970.
Eibl, H. & Unger, C. (1990). Hexadecylphosphocholine, a new and selective antitumor drug. Cancer Treatment Rev, 17: 233–242.
Eibl, H., Unger, C., Fleer, E. A. M., Kim, D. J., Berger, M. R. & Nagel, G. A. (1986). Hexadecylphosphocholine, a new antineoplastic agent: cytotoxic properties in leukaemic cells. J Cancer Res Clin Oncol, 111: S24
Fichtner, I., Zeisig, R., Naundorf, H., Jungmann, S., Arndt, D., Asongwe, G., Double, J. A. & Bibby, M. C. (1994). Antineoplastic activity of alkylphosphocholines (APC) in human breast carcinomas in vivo and in vitro; use of liposomes. Breast Cancer Res Treatment, 32: 269–279.
Goppelt-Struebe, M. & Winter, I. (1995). Effects of hexadecylphosphocholine on fatty acid metabolism: relation to cytotoxicity. Cancer Chemother Pharmacol, 35: 519–526.
Hilgard, P., Stekar, J., Voegeli, R., Engel, J., Schumacher, W., Eibl, H., Unger, C. & Berger, M. R. (1988). Characterization of the anti-tumor activity of hexadecylphosphocholine. Eur J Cancer Clin Oncol, 24: 1457–1461.
Hilgard, P., Harleman, J. H., Voegeli, R., Maurer, H. R., Echarti, C. & Unger, C. (1989). The antineoplastic activity of hexadecylphosphocholines. Proc Am Ass Cancer Res, 30: 2310
Hilgard, P., Klenner, T., Stekar, J. & Unger, C. (1993). Alkylphosphocholines: a new class of membrane-active anticancer agents. Cancer Chemother Pharm, 32: 90–95.
Planting, A. S. T., Stoter, G. & Verweij, J. (1993). Phase II study of daily oral miltefosine (hexadecylphosphocholine) in advanced colorectal cancer. Eur J Cancer, 29A: 518–519.
Unger, C. & Eibl, H. (1991). Hexadecylphosphocholine: preclinical and first clinical results of a new antitumor drug. Lipids, 26: 1412–1417.
Unger, C., Eibl, H., von Heyden, H. W. & Nagel, G. A. (1984). Reversible opening of the blood-brain-barrier for drugs transfer by short-chain alkyl glycerols. Proc Am Soc Clin Oncol, 20: 25
Unger, C., von Heyden, H. W., Breiser, A., Nagel, C. A. & Eibl, H. (1988). Etherlipids in the topical treatment of skin metastases. J Cancer Res Clin Oncol, 114: S40
Unger, C., Damenz, W., Fleer, E. A. M., Kim, D. J., Breiser, A., Hilgard, P., Engel, J., Nagel, G. & Eibl, H. (1989). Hexadecylphosphocholine, a new ether lipid analogue. Acta Oncologica, 28: 213–217.
Unger, C., Peukert, M., Sindermann, H., Hilgard, P., Nagel, G. & Eibl, H. (1990). Hexadecylphosphocholine in the topical treatment of skin metastases in breast cancer patients. Cancer Treatment Rev, 17: 243–246.
Unger, C., Sindermann, H., Peukert, M., Hilgard, P., Engel, J. & Eibl, H. (1992). Hexadecylphosphocholine in the topical treatment of skin metastases in breast cancer patients. Progr Exp Tumor Res, 34: 153–159.
Verweij, J., Planting, A., van der Burg, M. & Stoter, G. (1992). A dose-finding study of miltefosine (hexadecylphosphocholine) in patients with metastatic solid tumours. J Cancer Res Clin Oncol, 118: 606–608.
Verweij, J., Krzemieniecki, K., Kok, T., Poveda, A., van Pottelsberghe, C., van Glabbeke, M. & Mouridsen, H. (1993). Phase II study of miltefosine in advanced soft tissue sarcomas of the adult – an EORTC soft tissue and bone sarcoma group study. Eur J Cancer, 29A: 208–209.
Verweij, J., Gandia, D., Planting, A. S. T., Stoter, G. & Armand, J. P. (1993). Phase II study of oral miltefosine in patients with squamous cell head and neck cancer. Eur J Cancer, 29A: 778–779.
WHO (1997). WHO Handbook for Reporting Results of Cancer Treatment. World Health Organization: Geneva
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Terwogt, J., Mandjes, I., Sindermann, H. et al. Phase II trial of topically applied miltefosine solution in patients with skin-metastasized breast cancer. Br J Cancer 79, 1158–1161 (1999). https://doi.org/10.1038/sj.bjc.6690184
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690184
Keywords
This article is cited by
-
A Phase 2 study of perifosine in advanced or metastatic breast cancer
Breast Cancer Research and Treatment (2008)
-
Lysosomal and mitochondrial pathways in miltefosine-induced apoptosis in U937 cells
Apoptosis (2007)
-
Enhancement of antitumor activity of the anti-EGF receptor monoclonal antibody cetuximab/C225 by perifosine in PTEN-deficient cancer cells
Oncogene (2006)
-
The anti-leishmanial drug miltefosine causes insulin resistance in skeletal muscle cells in vitro
Diabetologia (2006)