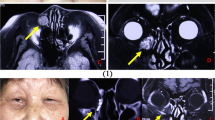
Abstract
We report a 71-year male with castration-resistant metastatic prostate cancer who was treated with weekly docetaxel for 12 weeks and developed significant eye irritation and dryness during treatment. Subsequently, the patient presented with a lower eyelid mass, which on excision was demonstrated to be a chalazion. Docetaxel induced Meibomian duct inflammation and blockage is the likely cause of this presentation in a patient with no history of eyelid masses in the past.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Dieras V, Chevallier B, Kerbrat P, Krakowski I, Roche H, Misset JL et al. A multicentre phase II study of docetaxel 75 mg m−2 as first-line chemotherapy for patients with advanced breast cancer: report of the Clinical Screening Group of the EORTC. European Organization for Research and Treatment of Cancer. Br J Cancer 1996; 74: 650–656.
Shepherd FA, Dancey J, Ramlau R, Mattson K, Gralla R, O’Rourke M et al. Prospective randomized trial of docetaxel versus best supportive care in patients with non-small-cell lung cancer previously treated with platinum-based chemotherapy. J Clin Oncol 2000; 18: 2095–2103.
Mavroudis D, Kourousis C, Androulakis N, Kalbakis K, Agelaki S, Kakolyris S et al. Frontline treatment of advanced gastric cancer with docetaxel and granulocyte colony-stimulating factor (G-CSF): a phase II trial. Am J Clin Oncol 2000; 23: 341–344.
Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004; 351: 1502–1512.
Valero V, Holmes FA, Walters RS, Theriault RL, Esparza L, Fraschini G et al. Phase II trial of docetaxel: a new, highly effective antineoplastic agent in the management of patients with anthracycline-resistant metastatic breast cancer. J Clin Oncol 1995; 13: 2886–2894.
Ferraresi V, Milella M, Vaccaro A, D'Ottavio AM, Papaldo P, Nistico C et al. Toxicity and activity of docetaxel in anthracycline-pretreated breast cancer patients: a phase II study. Am J Clin Oncol 2000; 23: 132–139.
Esmaeli B, Valero V, Ahmadi MA, Booser D . Canalicular stenosis secondary to docetaxel (taxotere): a newly recognized side effect. Ophthalmology 2001; 108: 994–995.
Perry HD, Serniuk RA . Conservative treatment of chalazia. Ophthalmology 1980; 87: 218–221.
Esmaeli B, Ahmadi MA, Rivera E, Valero V, Hutto T, Jackson DM et al. Docetaxel secretion in tears: association with lacrimal drainage obstruction. Arch Ophthalmol 2002; 120: 1180–1182.
Esmaeli B, Hortobagyi GN, Esteva FJ, Booser D, Ahmadi MA, Rivera E et al. Canalicular stenosis secondary to weekly versus every-3-weeks docetaxel in patients with metastatic breast cancer. Ophthalmology 2002; 109: 1188–1191.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest
No potential conflicts of interest to disclose.
Rights and permissions
About this article
Cite this article
Gupta, S., Silliman, C. & Trump, D. Docetaxel-induced Meibomian duct inflammation and blockage leading to chalazion formation. Prostate Cancer Prostatic Dis 10, 396–397 (2007). https://doi.org/10.1038/sj.pcan.4500939
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.pcan.4500939
Keywords
This article is cited by
-
Meibom-Drüsen
Der Ophthalmologe (2009)