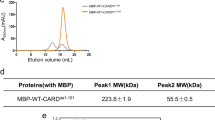
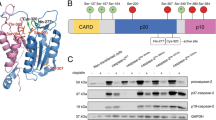
Abstract
Activation of procaspase-9, a key component of the apoptosis mechanism, requires the interaction of its caspase recruitment domain (CARD) with the CARD in the adaptor protein Apaf-1. Using nuclear magnetic resonance spectroscopy and mutagenesis we have determined the structure of the CARD from Apaf-1 and the residues important for binding the CARD in procaspase-9. Apaf-1's CARD contains seven short α-helices with the core six helices arranged in an antiparallel manner. Residues in helix 2 have a central role in mediating interaction with procaspase-9 CARD. This interaction surface is distinct from that proposed based on the structure of the CARD from RAIDD, but is coincident with that of the structurally similar FADD death effector domain and the Apaf-1 CARD interface identified by crystallographic studies.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by G Melino
Rights and permissions
About this article
Cite this article
Day, C., Dupont, C., Lackmann, M. et al. Solution structure and mutagenesis of the caspase recruitment domain (CARD) from Apaf-1. Cell Death Differ 6, 1125–1132 (1999). https://doi.org/10.1038/sj.cdd.4400584
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cdd.4400584
Keywords
This article is cited by
-
A chemical inhibitor of Apaf-1 exerts mitochondrioprotective functions and interferes with the intra-S-phase DNA damage checkpoint
Apoptosis (2009)
-
Programmed cell death in cardiac myocytes: strategies to maximize post-ischemic salvage
Heart Failure Reviews (2008)
-
Tissue distribution of Diablo/Smac revealed by monoclonal antibodies
Cell Death & Differentiation (2002)
-
Formation of the Apaf-1/cytochrome c complex precedes activation of caspase-9 during seizure-induced neuronal death
Cell Death & Differentiation (2001)
-
Caspase-8/FLICE functions as an executioner caspase in anticancer drug-induced apoptosis
Oncogene (2000)