Abstract
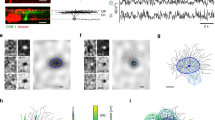
THE dendrites of mammalian pyramidal neurons contain a rich collection of active conductances that can support Na+ and Ca2+ action potentials (for a review see ref. 1). The presence, site of initiation, and direction of propagation of Na+ and Ca2+ action potentials are, however, controversial2, and seem to be sensitive to resting membrane potential, ionic composition, and degree of channel inactivation, and depend on the intensity and pattern of synaptic stimulation. This makes it difficult to extrapolate from in vitro experiments to the situation in the intact brain. Here we show that two-photon excitation laser scanning microscopy3 can penetrate the highly scattering tissue of the intact brain. We used this property to measure sensory stimulus-induced dendritic [Ca2+] dynamics of layer 2/3 pyramidal neurons of the rat primary vibrissa (Sm1) cortex in vivo. Simultaneous recordings of intracellular voltage and dendritic [Ca2+] dynamics during whisker stimulation or current injection showed increases in [Ca2+] only in coincidence with Na+ action potentials. The amplitude of these [Ca2+] transients at a given location was approximately proportional to the number of Na+ action potentials in a short burst. The amplitude for a given number of action potentials was greatest in the proximal apical dendrite and declined steeply with increasing distance from the soma, with little Ca2+ accumulation in the most distal branches, in layer 1. This suggests that widespread Ca2+ action potentials were not generated, and any significant [Ca2+] increase depends on somatically triggered Na+ action potentials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Yuste, R. & Tank, D. W. Neuron 16, 701–716 (1996).
Regehr, W. G. & Armstrong, C. M. Curr. Biol. 4, 436–439 (1994).
Denk, W., Strickler, J. H. & Webb, W. W. Science 248, 73–76 (1990).
Stuart, G. J. & Sakmann, B. Nature 367, 69–72 (1994).
Regehr, W. G., Connor, J. A. & Tank, D. W. Nature 341, 533–536 (1989).
Jaffe, D. B. et al. Nature 357, 244–246 (1992).
Magee, J. C. & Johnston, D. Science 268, 301–304 (1995).
Spruston, N., Schiller, Y., Stuart, G. & Sakmann, B. Science 268, 297–300 (1995).
Yuste, R. & Denk, W. Nature 375, 682–684 (1995).
Wong, R. K. S., Prince, D. A. & Basbaum, A. I. Proc. Natl Acad. Sci. USA 76, 986–990 (1979).
Kim, H. G. & Connors, B. W. J. Neurosci. 13, 5301–5311 (1993).
Yuste, R., Gutnick, M. J., Saar, D., Delaney, K. R. & Tank, D. W. Neuron 13, 23–43 (1994).
Andreasen, M. & Lambert, J. D. C. J. Physiol. (Lond.) 483, 421–441 (1995).
Turner, R. W., Meyers, E. R., Richardson, D. L. & Barker, J. L. J. Neurosci. 11, 2270–2280 (1991).
Kim, H. G., Beierlein, M. & Connors, B. W. J. Neurophysiol. 74, 1810–1814 (1995).
Buzsaki, G., Penttonen, M., Nadasdy, Z. & Bragin, A. Proc. Natl Acad. Sci. USA 93, 9921–9925 (1996).
Tsubokawa, H. & Ross, W. N. J. Neurophysiol. 76, 2896–2906 (1996).
Hirsch, J. A., Alonso, J.-M. & Reid, R. C. Nature 378, 612–616 (1995).
Denk, W. et al. J. Neurosci. Methods 54, 151–162 (1994).
Connors, B. W. & Gutnick, M. J. Trends Neurosci. 13, 99–104 (1990).
Carvell, G. E. & Simons, D. J. Brain Res. 448, 186–191 (1988).
Helmchen, F., Imoto, K. & Sakmann, B. Biophys. J. 70, 1069–1081 (1996).
Andreasen, M. & Hablitz, J. J. J. Neurophysiol. 69, 1966–1975 (1993).
Reuveni, I., Friedman, A., Amitai, Y. & Gutnick, M. J. J. Neurosci. 13, 4609–4621 (1993).
Gutfreund, Y., Yarom, Y. & Segev, I. J. Physiol. (Lond.) 483, 621–640 (1995).
Callaway, J. C. & Ross, W. N. J. Neurophysiol. 74, 1395–1403 (1995).
Bear, M. F. & Malenka, R. C. Curr. Opin. Neurobiol. 4, 389–399 (1994).
Kleinfeld, D. & Delaney, K. R. J. Comp. Neurol. 375, 89–108 (1996).
Carvell, G. E. & Simons, D. J. J. Neurosci. 10, 2638–2648 (1990).
Feller,,M. B., Delaney, K. R. & Tank, D. W. J. Neurophysiol. 76, 381–401 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Svoboda, K., Denk, W., Kleinfeld, D. et al. In vivo dendritic calcium dynamics in neocortical pyramidal neurons. Nature 385, 161–165 (1997). https://doi.org/10.1038/385161a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/385161a0
This article is cited by
-
Experimental test of the entanglement enhancement in two-photon fluorescence
Quantum Frontiers (2024)
-
Construction and use of an adaptive optics two-photon microscope with direct wavefront sensing
Nature Protocols (2023)
-
Minimally invasive longitudinal intravital imaging of cellular dynamics in intact long bone
Nature Protocols (2023)
-
Quantitative analyses of matrices, osteoblasts, and osteoclasts during bone remodeling using an in vitro system
Journal of Bone and Mineral Metabolism (2023)
-
Fast and sensitive GCaMP calcium indicators for imaging neural populations
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.