Abstract
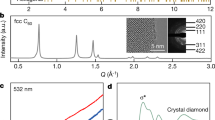
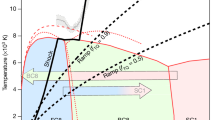
C6o MOLECULES are extremely stable, withstanding hydrostatic pressures of up to at least 20 GPa (ref. 1). It has been proposed that at high pressures they could form a solid harder than diamond2. On the other hand, electrical resistivity measurements3 have revealed the formation of an insulating phase above 20 GPa, which was attributed to the low-symmetry state found in X-ray diffraction studies under nonhydrostatic compression1. Here we report that rapid, nonhydrostatic compression of C60 to pressures of 20 ±5 GPa transforms it instantaneously into bulk polycrystal-line diamond at room temperature. Our measurements place a limit on the stability of the C60molecular phase under nonhydrostatic pressure. The high efficiency and fast kinetics at room temperature suggest the possibility of using this transformation for fabrication of industrial diamonds.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Duclos, S. J., Brister, K., Haddon, R. C., Kortan, A. R. & Thiel, F. A. Nature 351, 380–382 (1991).
Ruoff, R. S. & Ruoff, A. L. Nature 350, 663–664 (1991).
Núńez Regueiro, M., Monceau, P., Rassat, A., Bernier, P. & Zahab, A. Nature 354, 289–291 (1991).
Kraetschmer, W., Lamb, L. D., Fostiropoulos, L. D. & Huffman, D. R. Nature 347, 354–358 (1990).
Fleming, R. M. et al. Nature 352, 787–788 (1991).
O'Keeffe, M. Nature 352, 674 (1991).
Bundy, F. P. Physica A156, 169–178 (1989).
De Carli, P. S. & Jamieson, J. C. Science 133, 1821–1824 (1961).
Bundy, F. P., Hall, H. T., Strong, H. M. & Wentorf, R. H. Nature 176, 51–55 (1955).
Saito, S. & Oshiyama, A. Phys. Rev. Lett. 66, 2637 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Regueiro, M., Monceau, P. & Hodeau, JL. Crushing C60 to diamond at room temperature. Nature 355, 237–239 (1992). https://doi.org/10.1038/355237a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/355237a0
This article is cited by
-
Extending on-surface synthesis from 2D to 3D by cycloaddition with C60
Nature Communications (2023)
-
Carbon clusters formed from shocked benzene
Nature Communications (2021)
-
Diamond synthesis from carbon nanofibers at low temperature and low pressure
Scientific Reports (2015)
-
Polymerization of solid C60 under C 60 + cluster ion bombardment
Applied Physics A (2009)
-
Probing the structure of heterogeneous diluted materials by diffraction tomography
Nature Materials (2008)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.