Abstract
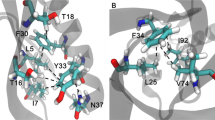
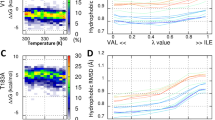
IT is rare for amino-acid substitutions on the surface of proteins to have large stabilizing or destabilizing effects1. Nevertheless, one substitution of this type, the Tyr 26 → Cys mutation in λ Cro, increases the melting temperature of the protein by 11°C and the stability by 2.2 kcal mol−1 (ref. 2). Here we show that the stability of Cro can be increased by many different amino-acid substitutions at position 26, with increasing stability showing a good correlation with decreasing side-chain hydrophobicity. As Tyr 26 is hyper-exposed to solvent in the Cro crystal structure3,4, we suggest that wild-type and variant proteins with other hydrophobic side chains at position 26 are destabilized as a result of a reverse hydrophobic effect caused by the side chain being more exposed to solvent in the native than in the denatured state.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Hecht, M. H., Sturtevant, J. M. & Sauer, R. T. Proc. natn. Acad. Sci. U.S.A. 81, 5685–5689 (1984).
Pakula, A. A. & Sauer, R. T. Proteins 5, 202–210 (1989).
Anderson, W. F., Ohlendorf, D. H., Takeda, Y. & Matthews, B. W. Nature 290, 754–758 (1981).
Ohlendorf, D. H., Anderson, W. F., Fisher, R. G., Takeda, Y. & Matthews, B. W. Nature 298, 718–723 (1982).
Reidhaar-Olson, J. F. & Sauer, R. T. Science 241, 53–57 (1988).
Fauchere, J.-L. & Pliska, V. Eur. J. Med. Chem. Chim. Ther. 18, 369–375 (1983).
Eisenberg, D. & McLachlan, A. D. Nature 319, 199–203 (1986).
Kauzmann, W. Adv. Prot. Chem. 14, 1–63 (1959).
Tanford, C. The Hydrophobic Effect 2nd Edn (Wiley–lnterscience, New York, 1980).
Lee, B. K. & Richards, F. M. J. molec. Biol. 55, 379–400 (1971).
Nicholson, H., Becktel, W. J. & Matthews, B. W. Nature 336, 651–656 (1988).
Shortle, D. & Meeker, A. K. Biochemistry 28, 936–944 (1989).
Ptitsyn, O. B. J. Prot. Chem. 6, 273–293 (1987).
Bashford, D., Chothia, C. & Lesk, A. M. J. molec. Biol. 196, 199–216 (1987).
Reidhaar-Olson, J. F. & Sauer, R. T. Proteins (in the press).
Bowie, J. U. & Sauer, R. T. J. biol. Chem. 264, 7596–7602 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pakula, A., Sauer, R. Reverse hydrophobic effects relieved by amino-acid substitutions at a protein surface. Nature 344, 363–364 (1990). https://doi.org/10.1038/344363a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/344363a0
This article is cited by
-
A hydrophobic ratchet entrenches molecular complexes
Nature (2020)
-
Loss of a conserved salt bridge in bacterial glycosyl hydrolase BgIM-G1 improves substrate binding in temperate environments
Communications Biology (2018)
-
Rational stabilization of complex proteins: a divide and combine approach
Scientific Reports (2015)
-
ProtSA: a web application for calculating sequence specific protein solvent accessibilities in the unfolded ensemble
BMC Bioinformatics (2009)
-
Surface areas of unfolded proteins
Nature (1990)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.