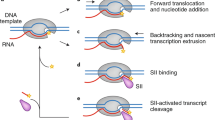
Abstract
RAP30/74 is a heteromeric general transcription initiation factor which binds to RNA polymerase II. Here we report that preparations of RAP30/74 contain an ATP-dependent DNA helicase whose probable function is to melt the DNA at transcrip-tional start sites. The sequence of the RAP30 subunit of RAP30/74 indicates that RAP30 may be distantly related to bacterial sigma factors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bunick, D., Zandomeni, R., Ackerman, S. & Weinmann, R. Cell 29, 877–886 (1982).
Davison, B. L., Egly, J-M., Mulvihill, E. R. & Chambon, P. Nature 301, 630–686 (1983).
Parker, C. S. & Topol, J. Cell 36, 357–369 (1984).
Nakajima, N., Horikoshi, M. & Roeder, R. G. Molec. cell. Biol. 8, 4028–4040 (1988).
Buratowski, S., Hahn, S., Sharp, P. A. & Guarante, L. Nature 334, 37–42 (1988).
Cavallini, B. et al. Nature 334, 77–80 (1988).
Kadonaga, J. T., Jones, K. A. & Tjian, R. Trends Biochem. 11, 20–23 (1986).
Matsui, T., Segall, J., Weil, P. A. & Roeder, R. G. J. biol. Chem. 255, 11992–11996 (1980).
Samuels, M., Fire, A. & Sharp, P. A. J. biol. Chem. 257, 14419–14427 (1982).
Flores, O., Maldonado, E., Burton, Z. F., Greenblatt, J. & Reinberg, D. J. biol. Chem. 263, 10812–10816 (1988).
Sopta, M., Carthew, R. W. & Greenblatt, J. J. biol. Chem. 260, 10353–10361 (1985).
Reinberg, D. & Roeder, R. G. J. biol. Chem. 262, 3310–3321 (1987).
Zheng, X-M., Moncollin, V., Egly, J-M. & Chambon, P. Cell 50, 361–368 (1987).
Fire, A., Samuels, M. & Sharp, P. A. J. biol. Chem. 259, 2509–2516 (1984).
Burton, Z. F., Ortolan, L. G. & Greenblatt, J. EMBO J. 5, 2923–2930 (1986).
Burton, Z. F., Killeen, M., Sopta, M., Ortolan, L. G. & Greenblatt, J. Molec. cell. Biol. 8, 1602–1613 (1988).
VanDyke, M. W., Roeder, R. G. & Sawadogo, M. Science 241, 1335–1338 (1988).
Buratowski, S., Hahn, S., Guarente, L. & Sharp, P. A. Cell 56, 549–561 (1989).
Conaway, J. W. & Conaway, R. C. J. biol. Chem. 264, 2357–2362 (1989).
Stahl, H., Droge, P. & Knippers, R. EMBO J. 5, 1939–1944 (1986).
Matson, S. W. J. biol. Chem. 26, 10169–10175 (1986).
Goetz, S. G., Dean, F. B., Hurwitz, J. & Matson, S. J. biol. Chem. 263, 383–392 (1988).
Geider, K. & Hoffman-Berling, H. A. Rev. Biochem. 50, 233–260 (1981).
Sawadogo, M. & Roeder, R. G. J. biol. Chem. 259, 5321–5326 (1984).
Hawley, D. K. & Roeder, R. G. J. biol. Chem. 260, 8163–8172 (1985).
Young, R. A. & Davis, R. W. Proc. natn. Acad. Sci. U.S.A. 80, 1194–1198 (1983).
Kozak, M. Microbiol. Rev. 47, 1–45 (1983).
Moller, W. & Amons, R. FEBS Lett. 186, 1–7 (1985).
Pabo, C. O. & Sauer, R. T. A. Rev. Biochem. 53, 293–321 (1984).
Miller, J., McLachlan, A. D. & Klug, A. EMBO J. 4, 1609–1614 (1985).
Mitchell, P. J. & Tjian, R. Science 245, 371–378 (1989).
Gonzalez, G. A. et al. Nature 337, 749–752 (1989).
Linder, P. et al. Nature 337, 121–122 (1989).
Ishii, S. et al. Nucleic Acids Res. 12, 3333–3342 (1984).
Stragier, P., Parsot, C. & Bouvier, J. FEBS Lett. 187, 11–15 (1985).
Gribskov, M. & Burgess, R. R. Nucleic Acids Res. 14, 6745–6763 (1986).
Hellmann, J. D. & Chamberlin, M. J. A. Rev. Biochem. 57, 839–872 (1988).
Strickland, M. S., Thompson, N. E. & Burgess, R. R. Biochemistry 27, 5755–5762 (1988).
Hu, J. C. & Gross, C. A. Molec. Gen. Genet. 191, 492–498 (1983).
Dayhoff, M. O., Barker, W. C. & Hunt, L. T. Meth. Enzym. 91, 524–545 (1983).
Chamberlin, M. J. A. Rev. Biochem. 43, 721–775 (1974).
Travers, A. A. & Burgess, R. R. Nature 222, 537–540 (1969).
Smale, S. T. & Baltimore, D. Cell 57, 103–113 (1989).
Hawley, D. K. & McClure, R. W. J. molec. Biol. 157, 493–525 (1982).
Ninfa, A. J., Reitzer, L. J. & Magasanik, B. Cell 50, 1039–1046 (1987).
Shapiro, D. J., Sharp, P. A., Wahli, W. W. & Keller, M. J. DNA, in the press.
Ausubel, F. M. et al. (eds) Current Protocols in Molecular Biology Vol. 1 (Wiley, New York, 1987).
Sadowski, I., Stone, J. C. & Pawson, T. Molec. cell. Biol. 6, 4396–4408 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sopta, M., Burton, Z. & Greenblatt, J. Structure and associated DNA-helicase activity of a general transcription initiation factor that binds to RNA polymerase II. Nature 341, 410–414 (1989). https://doi.org/10.1038/341410a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/341410a0
This article is cited by
-
Variants in the 3' UTR of General Transcription Factor IIF, polypeptide 2 affect female calving efficiency in Japanese Black cattle
BMC Genetics (2013)
-
Architecture of the RNA polymerase II–TFIIF complex revealed by cross-linking and mass spectrometry
The EMBO Journal (2010)
-
Basal transcription machinery: role in regulation of stress response in eukaryotes
Journal of Biosciences (2007)
-
Structure and mechanism of the RNA polymerase II transcription machinery
Nature Structural & Molecular Biology (2004)
-
Interaction with general transcription factor IIF (TFIIF) is required for the suppression of activated transcription by RPB5-mediating protein (RMP)
Cell Research (2003)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.