Abstract
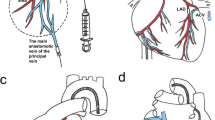
While a number of virus-based delivery schemes have been developed for myocardial gene transfer, no technique has proven capable of modifying a majority of cardiac myocytes rapidly and homogeneously in the in vivo rat model, and most schemes result in significant infection of the liver and other organs. However, adenoviral delivery to the excised heart during retrograde perfusion can produce 67–92% efficient gene transfer. In this study, we adapt this isolation/perfusion scheme to the heart in vivo. We isolated the heart in vivo by simultaneously clamping all vessels to/from the heart. The heart was then continuously retrograde perfused through a catheter positioned in the aortic root. A second catheter in the right ventricle provided a path for efflux. After perfusing the heart for 7.5 min with calcium-free Tyrode solution followed by 90 s no-flow viral exposure (AdV.cmv.LacZ; 1012 viral particles/ml), gene transfer efficiency was 60% compared to 5% by a conventional cross-clamp technique. Infection of peripheral organs was dramatically reduced. Given the prevalence of the rat in so many models of heart disease, this enhancement of infection represents an advancement in viral-based delivery of exogenous genes to heart for the study of gene therapy in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Svensson EC et al. Efficient and stable transduction of cardiomyocytes after intramyocardial injection or intracoronary perfusion with recombinant adeno-associated virus vectors. Circulation 1999; 99: 201–205.
Shah AS et al. Intracoronary adenovirus-mediated delivery and overexpression of the β-adrenergic receptor in the heart: prospects for molecular ventricular assistance. Circulation 2000; 101: 408–414.
Ikeda Y et al. Restoration of deficient membrane proteins in the cardiomyopathic hamster by in vivo cardiac gene transfer. Circulation 2002; 105: 502–508.
Hajjar RJ et al. Modulation of ventricular function through gene transfer in vivo. Proc Natl Acad Sci USA 1998; 95: 5251–5256.
Bridges CR et al. Global cardiac-specific transgene expression using cardiopulmonary bypass with cardiac isolation. Ann Thorac Surg. 2002; 73: 1939–1946.
Bekeredjian R et al. Ultrasound-targeted microbubble destruction can repeatedly direct highly specific plasmid expression to the heart. Circulation 2003; 108: 1022–1026.
Champion HC et al. Robust adenoviral and adeno-associated viral gene transfer to the in vivo murine heart. Circulation 2003; 108: 2790–2797.
Iwatate M et al. In vivo high-efficiency transcoronary gene delivery and Cre-LoxP gene switching in the adult mouse heart. Gene Therapy 2003; 10: 1814–1820.
Donahue JK et al. Ultrarapid, highly efficient viral gene transfer to the heart. Proc Natl Acad Sci. USA 1997; 94: 4664–4669.
Donahue JK et al. Acceleration of widespread adenoviral gene transfer to intact rabbit hearts by coronary perfusion with low calcium and serotonin. Gene Therapy 1998; 5: 630–634.
Logeart D et al. Highly efficient adenovirus-mediated gene transfer to cardiac myocytes after single-pass coronary delivery. Hum Gene Ther 2000; 11: 1015–1022.
Wright MJ, Wightman LML, Latchman DS, Marber MS . In vivo myocardial gene transfer: optimization and evaluation of intracoronary gene delivery in vivo. Gene Therapy 2001; 8: 1833–1839.
Bett AJ, Prevec L, Graham FL . Packaging capacity and stability of human adenovirus type 5 vectors. J Virol 1993; 67: 5911–5921.
Fleury S et al. Multiply attenuated, self-inactivating lentiviral vectors efficiently deliver and express genes for extended periods of time in adult rat cardiomyocytes in vivo. Circulation 2003; 107: 2375–2382.
DelMonte F, Hajjar RJ . Efficient viral gene transfer to rodent hearts in vivo. In: Metzger JM (ed). Methods in Molecular Biology, Vol 219: Cardiac Cell and Gene Transfer. Humana Press Inc.: Totowa NJ, 2002, pp 179–193.
Emani SM et al. Catheter-based intracoronary myocardial adenoviral gene delivery: importance of intraluminal seal and infusion flow rate. Mol Ther 2003; 8: 306–313.
O'Donnell JM et al. Tight control of exogenous SERCA expression is required to obtain acceleration of calcium transients with minimal cytotoxic effects in cardiac myocytes. Circ Res 2001; 88: 415–421.
Acknowledgements
We thank Drs David Geenen and Steven Pogwizd (UIC, Cardiology) for helpful discussion. This study was supported by grants from the American Heart Association (O'Donnell; 0230099N) and the National Heart, Lung, and Blood Institute, NIH (Lewandowski; RO1HL 62702, RO1HL 56178, R37HL49244).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
O'Donnell, J., Lewandowski, E. Efficient, cardiac-specific adenoviral gene transfer in rat heart by isolated retrograde perfusion in vivo. Gene Ther 12, 958–964 (2005). https://doi.org/10.1038/sj.gt.3302477
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302477
Keywords
This article is cited by
-
Cardiac gene therapy: are we there yet?
Gene Therapy (2016)
-
Percutaneous Approaches for Efficient Cardiac Gene Delivery
Journal of Cardiovascular Translational Research (2013)
-
Locoregional intravascular viral therapy of cancer: precision guidance for Paris's arrow?
Gene Therapy (2010)