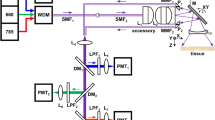
Abstract
Current methods for detecting successful gene transfer to airway epithelia involve obtaining a sample of the target tissue. This may affect the longevity of expression of the transgene under evaluation. We describe a laser fluorescence bronchoscopic system that can detect the expression of the fluorescent protein, green fluorescence protein (GFP), in the airway of monkeys that have been transfected with adenovirus, without the need for obtaining tissue. This technique will have applications in pre-clinical and clinical studies of gene transfer to airway epithelia and other surface epithelia accessible by endoscopy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Curiel DT, Pilewski JM, Albelda SM . Gene therapy approaches for inherited and acquired lung diseases Am J Respir Cell Mol Biol 1996 14: 1–18
Robinson CB . Is DNA destiny? A cure for cystic fibrosis Clin Chest Med 1998 19: 527–534
Knowles MR et al. A controlled study of adenoviral-vector-mediated gene transfer in the nasal epithelium of patients with cystic fibrosis N Engl J Med 1995 333: 823–831
Zabner J et al. Adenovirus-mediated gene transfer transiently corrects the chloride transport defect in nasal epithelia of patients with cystic fibrosis Cell 1993 75: 207–216
Crystal RG et al. Administration of an adenovirus containing the human CFTR cDNA to the respiratory tract of individuals with cystic fibrosis Nat Genet 1994 8: 42–51
Alton EW et al. Cationic lipid-mediated CFTR gene transfer to the lungs and nose of patients with cystic fibrosis: a double-blind placebo-controlled trial Lancet 1999 353: 947–954
Knowles MR, Paradiso AM, Boucher RC . In vivo nasal potential difference: techniques and protocols for assessing efficacy of gene transfer in cystic fibrosis Hum Gene Ther 1995 6: 445–455
Middleton PG, Geddes DM, Alton EW . Protocols for in vivo measurement of the ion transport defects in cystic fibrosis nasal epithelium Eur Respir J 1994 7: 2050–2056
Roth JA et al. Retrovirus-mediated wild-type p53 gene transfer to tumors of patients with lung cancer Nat Med 1996 2: 985–991
Weill D et al. Adenoviral-mediated p53 gene transfer to non-small cell lung cancer through endobronchial injection Chest 2000 118: 966–970
Tursz T et al. Phase I study of a recombinant adenovirus-mediated gene transfer in lung cancer patients J Natl Cancer Inst 1996 88: 1857–1863
Frey BM et al. High-efficiency gene transfer into ex vivo expanded human hematopoietic progenitors and precursor cells by adenovirus vectors Blood 1998 91: 2781–2792
Chalfie M et al. Green fluorescent protein as a marker for gene expression Science 1994 263: 802–805
Niswender KD et al. Quantitative imaging of green fluorescent protein in cultured cells: comparison of microscopic techniques, use in fusion proteins and detection limits J Microsc 1995 180: 109–116
Persons DA et al. Use of the green fluorescent protein as a marker to identify and track genetically modified hematopoietic cells Nat Med 1998 4: 1201–1205
Flotte TR et al. A fluorescence video-endoscopy technique for detection of gene transfer and expression Gene Therapy 1998 5: 166–173
Kusunoki Y et al. Early detection of lung cancer with laser-induced fluorescence endoscopy and spectrofluorometry Chest 2000 118: 1776–1782
Yu Y et al. Quantification of target gene expression by imaging reporter gene expression in living animals Nat Med 2000 6: 933–937
Wu JC et al. Noninvasive optical imaging of firefly luciferase reporter gene expression in skeletal muscles of living mice Mol Ther 2001 4: 297–306
Walters RW et al. Basolateral localization of fiber receptors limits adenovirus infection from the apical surface of airway epithelia J Biol Chem 1999 274: 10219–10226
Pickles RJ et al. Retargeting the coxsackievirus and adenovirus receptor to the apical surface of polarized epithelial cells reveals the glycocalyx as a barrier to adenovirus-mediated gene transfer J Virol 2000 74: 6050–6057
Zabner J et al. Repeat administration of an adenovirus vector encoding cystic fibrosis transmembrane conductance regulator to the nasal epithelium of patients with cystic fibrosis J Clin Invest 1996 97: 1504–1511
Norris PS, Haas M . A fluorescent p53GFP fusion protein facilitates its detection in mammalian cells while retaining the properties of wild-type p53 Oncogene 1997 15: 2241–2247
Gunji Y et al. Gene therapy for cancer Surg Today 2000 30: 967–973
Kollen WJ et al. High-efficiency transfer of cystic fibrosis transmembrane conductance regulator cDNA into cystic fibrosis airway cells in culture using lactosylated polylysine as a vector Hum Gene Ther 1999 10: 615–622
Trapnell BC et al. Expression of the cystic fibrosis transmembrane conductance regulator gene in the respiratory tract of normal individuals and individuals with cystic fibrosis Proc Natl Acad Sci USA 1991 88: 6565–6569
Zabner J et al. Adenovirus-mediated gene transfer to ciliated airway epithelia requires prolonged incubation time J Virol 1996 70: 6994–7003
Welsh MJ et al. Cystic fibrosis gene therapy using an adenovirus vector: in vivo safety and efficacy in nasal epithelium Hum Gene Ther 1994 5: 209–219
Walters RW et al. Apical localization of the coxsackie-adenovirus receptor by glycosyl-phosphatidylinositol modification is sufficient for adenovirus-mediated gene transfer through the apical surface of human airway epithelia J Virol 2001 75: 7703–7711
Acknowledgements
We thank Pary Weber, Phil Karp, Lynn Kuchemann, Janice Launspach, Paul Montague, Tatiana Rokhlina, and Rosanna Smith for excellent assistance. We thank Dan Vermeer and Paola Drapkin for their help in developing the rabbit tracheal abrasion technique. We especially appreciate the help of Iowa Donor Network (IDN) and Tissue Transformation Technologies (T3) for acquisition of human lung tissue. We appreciate the support of the University of Iowa Gene Transfer Vector Core (supported by the Roy J Carver Charitable Trust, the NHLBI, CFF, and NIDDK-DK54759); In Vitro Cell Models Core (supported by the NIHLBI, CFF, and NIDDK-DK54759); and the Gene Transfer Morphology Core (supported by the NIDDK-DK54759). This work was supported by the National Heart, Lung and Blood Institute HL58340, the Cystic Fibrosis Foundation, and the Gene Therapy Center of the University of Iowa.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rooney, C., Suter, M., McLennan, G. et al. Laser fluorescence bronchoscopy for detection of fluorescent reporter genes in airway epithelia. Gene Ther 9, 1639–1644 (2002). https://doi.org/10.1038/sj.gt.3301825
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301825