Abstract
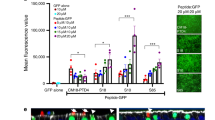
To identify the intracellular barriers to efficient gene transfer, we studied the intracellular trafficking of biotinylated plasmid DNA complexed with either fluorescein-conjugated lactosylated or mannosylated polylysine by confocal microscopy. Both are known to be taken up by cystic fibrosis airway epithelial cells (ΣCFTE29o− cells), but their gene transfer efficiencies differ markedly: lactosylated polylysine is the most efficient glycosylated polylysine while mannosylated polylysine is quite inefficient for gene transfer. Mannosylated complexes appeared to stay longer in endosomes labeled by anti-transferrin receptor antibody than lactosylated complexes (from 30 min to 3 h and from 10 min to 30 min, respectively). At 24 h, higher percentages of mannosylated than lactosylated complexes were localized inside lysosomes labeled by anti-LAMP-1 antibody (41.8 ± 6.6% versus 19.8 ± 5.2%, respectively, P < 0.05). Between 30 min and 8 h, complexes reached the nuclei labeled by anti-lamin A/C antibody and no difference was observed between the nuclear amounts of mannosylated and lactosylated complexes. However, as analyzed by a nuclease S1 transcription assay, initiation of transcription was prevented when plasmid DNA was complexed to mannosylated polylysine. Our results indicate that the major limiting steps for mannosylated versus lactosylated polylysine transfer of plasmid DNA are delayed exit from endosomes, high accumulation in lysosomes and limited transcription of the complexed plasmid DNA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Pouton CW, Seymour LW . Key issues in non-viral gene delivery Adv Drug Delivery Rev 1998 34: 3–19
Luo D, Saltzman WM . Synthetic DNA delivery systems Nat Biotechnol 2000 18: 33–37
Midoux P et al. Specific gene transfer mediated by lactosylated poly-L-lysine into hepatoma cells Nucleic Acids Res 1993 21: 871–878
Perales JC et al. Gene transfer in vivo: sustained expression and regulation of genes introduced into the liver by receptor-targeted uptake Proc Natl Acad Sci USA 1994 91: 4086–4090
Wadhwa MS, Knoell DL, Young AP, Rice KG . Targeted gene delivery with a low molecular weight glycopeptide carrier Bioconj Chem 1995 6: 283–291
Kollen WJW et al. Gluconoylated and glycosylated polylysines as vectors for gene transfer into cystic fibrosis airway epithelial cells Hum Gene Ther 1996 7: 1577–1586
Fajac I, Briand P, Monsigny M, Midoux P . Sugar-mediated uptake of glycosylated polylysines and gene transfer into normal and cystic fibrosis airway epithelial cells Hum Gene Ther 1999 10: 395–406
Allo JC et al. Efficient gene transfer into human normal and cystic fibrosis tracheal gland serous cells with synthetic vectors Am J Respir Cell Mol Biol 2000 22: 166–175
Heuser JE, Anderson RG . Hypertonic media inhibit receptor-mediated endocytosis by blocking clathrin-coated pit formation J Cell Biol 1989 108: 389–400
Pratten MK, Lloyd JB . Pinocytosis and phagocytosis: the effect of size of a particulate substrate on its mode of capture by rat peritoneal macrophages cultured in vitro Biochim Biophys Acta 1986 881: 307–313
Wu GY, Wu CH . Receptor-mediated in vitro gene transformation by a soluble DNA carrier system J Biol Chem 1987 262: 4429–4432
Curiel DT, Agarwal S, Wagner E, Cotten M . Adenovirus enhancement of transferrin-polylysine-mediated gene delivery Proc Natl Acad Sci USA 1991 88: 8850–8854
Colin M et al. Cell delivery, intracellular trafficking and expression of an integrin-mediated gene transfer vector in tracheal epithelial cells Gene Therapy 2000 7: 139–152
Zabner J et al. Cellular and molecular barriers to gene transfer by cationic lipid J Biol Chem 1995 270: 18997–19007
Zhou X, Huang L . DNA transfection mediated by cationic liposomes containing lipopolylysine: characterization and mechanism of action Biochim Biophys Acta 1994 1189: 195–203
Zenke M et al. Receptor-mediated endocytosis of transferrin-polycation conjugates: an efficient way to introduce DNA into hematopoietic cells Proc Natl Acad Sci USA 1990 87: 3655–3659
Erbacher P, Roche AC, Monsigny M, Midoux P . Putative role of chloroquine in gene transfer into a human hepatoma cell line by DNA/lactosylated polylysine complexes Exp Cell Res 1996 225: 186–196
Labat-Moleur F et al. An electron microscopy study into the mechanism of gene transfer with lipopolyamines Gene Therapy 1996 3: 1010–1017
Pollard H et al. Polyethylenimine but not cationic lipids promotes transgene delivery to the nucleus in mammalian cells J Biol Chem 1998 273: 7507–7511
Godbey WT, Wu KK, Mikos AG . Tracking the intracellular path of poly(ethylenimine)/DNA complexes for gene delivery Proc Natl Acad Sci USA 1999 96: 5177–5181
Fisher KD et al. A versatile system for receptor-mediated gene delivery permits increased entry of DNA into target cells, enhanced delivery to the nucleus and elevated rates of transgene expression Gene Therapy 2000 7: 1337–1343
Klink DT, Chao S, Glick MC, Scanlin TF . Nuclear translocation of lactosylated poly-L-lysine/cDNA complex in cystic fibrosis airway epithelial cells Mol Ther 2001 3: 831–841
Zauner W et al. Differential behaviour of lipid based and polycation based gene transfer systems in transfecting primary human fibroblasts: a potential role of polylysine in nuclear transport Biochim Biophys Acta 1999 1428: 57–67
Chan CK, Jans DA . Enhancement of polylysine-mediated transferrinfection by nuclear localization sequences: polylysine does not function as a nuclear localization sequence Hum Gene Ther 1999 10: 1695–1702
Duverger E et al. Nuclear import of glycoconjugates is distinct from the classical NLS pathway J Cell Sci 1995 108: 1325–1332
Andrulis ED, Neiman AM, Zappulla DC, Sternglanz R . Perinuclear localization of chromatin facilitates transcriptional silencing Nature 1998 394: 592–595
Kunzelmann K et al. An immortalized cystic fibrosis tracheal epithelial cell line homozygous for the △F508 CFTR mutation Am J Respir Cell Mol Biol 1993 8: 522–529
Derrien D et al. Muramyl dipeptide bound to poly-L-lysine substituted with mannose and gluconoyl residues as macrophage activators Glycoconj J 1989 6: 241–255
Monsigny M et al. Characterization and biological implications of membrane lectins in tumor, lymphoid and myeloid cells Biochimie 1988 70: 1633–1649
Erbacher P, Roche AC, Monsigny M, Midoux P . Glycosylated polylysine/DNA complexes: gene transfer efficiency in relation with the size and the sugar substitution level of glycosylated polylysines and with the plasmid size Bioconjug Chem 1995 6: 401–410
de Wet JR et al. Firefly luciferase gene: structure and expression in mammalian cells Mol Cell Biol 1987 7: 725–737
Smith PK et al. Measurement of protein using bicinchoninic acid Anal Biochem 1985 150: 76–85
Hill HD, Straka JG . Protein determination using bicinchoninic acid in the presence of sulfhydryl reagents Anal Biochem 1988 170: 203–208
Hill HD, Straka JG . Protein determination using bicinchoninic acid in the presence of sulfhydryl reagents Sambrook J, Fritsch E, Maniatis T. Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, 1989.
Acknowledgements
We are grateful to DC Gruenert for his gift of ΣCFTE29o− cells; P Bouchard for preparing the sugar derivatives; I Bouchaert (Service Commun de Microscopie Confocale, ICGM) for her help in confocal analysis; JM Egly for welcoming us in his laboratory and his help on transcription studies; P Midoux for technical advice and M Monsigny for helpful discussions. YA was funded by the Association Claude Bernard. This research was supported by the Association Vaincre la Mucoviscidose, The Chancellerie des Universités de Paris and the INSERM (CReS 003).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Grosse, S., Tremeau-Bravard, A., Aron, Y. et al. Intracellular rate-limiting steps of gene transfer using glycosylated polylysines in cystic fibrosis airway epithelial cells. Gene Ther 9, 1000–1007 (2002). https://doi.org/10.1038/sj.gt.3301768
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301768