Abstract
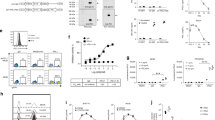
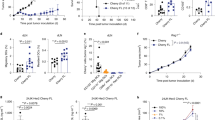
Genetic modification of tumor cells with the gene for the B7.1 or with the genes for cytokines results in increased tumor cell immunogenicity. In the work reported here, immunization of naive animals with either B7.1 or γ-IFN gene-modified MCA106 tumor cells effectively protects the host from subsequent challenge with parental tumor. The same treatment fails to induce regression of established tumors, although tumor-specific CTL are generated in the tumor-bearing animals. In contrast, a large tumor burden of the MCA106 fibrosarcoma can be successfully eliminated by treatment with MCA106 tumor cells cotransduced with the B7.1 and γ-IFN genes. Antitumor immunity induced by the cotransductants is primarily dependent on CD8+ T cells and partly on CD4+ T cells and NK cells, and the enhanced therapeutic effect may be attributed to the in vivo increase of CTL precursors following treatment. The γ-IFN and B7.1 genes must be expressed on the same tumor cell for optimal therapeutic effect. Our results suggest that tumor vaccines with a potent immunoprotective effect do not necessarily have therapeutic potential and that weakly immunogenic tumors may be rendered highly immunogenic by cotransfection with the genes for B7.1 and γ-IFN.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Mueller DL, Jenkins MK, Schwartz RH . Clonal expansion versus functional clonal inactivation: a costimulatory signaling pathway determines the outcome of T cell antigen receptor occupancy Annu Rev Immunol 1989 7: 445–480
Linsley PS, Ledberter JA . The role of the CD28 receptor during T cell responses to antigen Annu Rev Immunol 1993 11: 191–212
Schwartz RH . A cell culture model for T lymphocyte clonal anergy Science 1990 248: 1349–1356
Sharpe AH . Analysis of lymphocyte costimulation in vivo using transgenic and ‘knockout’ mice Curr Opin Immunol 1995 7: 389–395
Schwartz RH . Costimulation of T lymphocytes: the role of CD28, CTLA4, and B7/BB1 in interleukin-2 production and immunotherapy Cell 1992 71: 1065–1068
Chen L, Linsley PS, Hellstrom KE . Costimulation of T cells for tumor immunity Immunol Today 1993 14: 483–486
Chen L et al. Costimulation of antitumor immunity by the B7 counter receptor for the T lymphocyte molecules CD28 and CTLA-4 Cell 1992 71: 1093–1102
Townsend SE, Allison JP . Tumor rejection after direct costimulation of CD8+ T cells by B7-transfected melanoma cells Science 1993 259: 368–370
Ramarathinam L, Castle M, Wu Y, Liu Y . T cell costimulation by B7/BB1 induces CD8 T cell-dependent tumor rejection: an important role of B7/BB1 in the induction, recruitment, and effector function of antitumor T cells J Exp Med 1994 179: 1205–1214
Baskar S et al. Constitutive expression of B7 restores immunogenicity of tumor cells expressing truncated MHC class IImolecules Proc Natl Acad Sci USA 1993 90: 5687–5690
Li Y et al. Costimulation of tumor-reactive CD4+ and CD8+ T lymphocytes by B7, a natural ligand for CD28, can be used to treat established mouse melanoma J Immunol 1994 153: 421–428
Chen L et al. Tumor immunogenicity determines the effect of B7 costimulation on T cell-mediated tumor immunity J Exp Med 1994 179: 523–532
Yang G, Hellstrom KE, Hellstrom I, Chen L . Antitumor immunity elicited by tumor cells transfected with B7–2, a second ligand for CD28/CTLA-4 costimulatory molecules J Immunol 1995 154: 2794–2800
Mukherji D, Chakraborty NG . Immunobiology and immuno-therapy of melanoma. Curr Opin Oncol 1995; 7: 175–184
Seliger B, Maeurer MJ, Ferrone S . TAP off-tumors on Immunol Today 1997 18: 292–299
Gattido F, Ruiz-Cabello F . MHC expression of human tumors: its relevance for local tumor growth and metastasis SeminCancer Biol 1991 2: 3–10
Ruiter D, Mattijssen V, Brooecker EB, Ferrone S . MHC antigens in human melanomas Semin Cancer Biol 1991 2: 35–45
Ostrand-Rosenberg S, Cole GA, Nishimura MI, Clements VK . Transfection and expression of syngeneic H-2 genes does not reduce malignancy of H-2 negative teratocarcinoma cells in the autologous host Cell Immunol 1990 128: 152–164
Mandelboim O, Feldman M, Eisenbach L . H-2K double transfectants of tumor cells as antimetastatic cellular vaccines inheterozygous recipients. Implications for the T cell repertoire J Immunol 1992 148: 3666–3673
Xu F, Carlos T, Khokha R, Gorelik E . Reduction of metastatic and adhesion properties and up-regulation of TIMP-1 in BL6 melanoma cells transfected with MHC class I genes Proc Am Assoc Cancer Res 1997 38: 166 (Abstr. 141)
Ostrand-Rosenberg S, Clements VK, Armstrong T, Pulaski B . Immunotherapy with class II-transfected tumor cells. Proc Am Assoc Cancer Res. 1997; 38: 346 (Abstr 2318)
Yim JH, Marti W, Norton JA, Tsung K . Expression and antitumor effect of I-Ak by gene transfer into Sal sarcoma cells using a non-cytolytic recombinant vaccinia virus Proc Am Assoc Cancer Res 1997 38: 12 (Abstr. 79)
Dar MM et al. Immunological memory induced by genetically transduced tumor cells Ann Surg Oncol 1996 3: 247–254
Raes G et al. Immunogenization of a murine T-cell lymphoma via transfection with interferon-gamma Leukemia 1995 9 (Suppl.1): S121–S127
Lurquin C et al. Structure of the gene of tum- transplantation antigen p91A: the mutated exon encodes a peptide recognized with Ld by cytolytic T cells Cell 1989 58: 293–303
Hellstrom KE, Hellstrom I . Principles of tumor immunity: tumor antigens. In: De Vita VT Jr, Hellman S, Rosenberg SA (eds). Biologic Therapy of Cancer JB Lippincott Co: Philadelphia, 1991, p35
Houghton AN . Cancer antigens: immune recognition of self and altered self J Exp Med 1994 180: 1–4
Boon T et al. Tumor antigens recognized by T lymphocytes. Annu Rev Immunol 1994; 12: 337–365
Shu S, Rosenberg SA . Adoptive immunotherapy of newly induced murine sarcomas Cancer Res 1985 45: 1657–1662
Liu Y, Janeway C . Cells that present both specific ligand and costimulatory activity are the most efficient inducers of clonal expansion of normal CD4 T cells. Proc Natl Acad Sci USA 1992; 89: 3845–3849
Baskar S et al. Major histocompatibility complex class II+B7-1+ tumor cells are potent vaccines for stimulating tumor rejection in tumor-bearing mice J Exp Med 1995 181: 619–629
Schoenberger S et al. Efficient direct priming of tumor-specific cytotoxic T lymphocytes in vivo by an engineered APC Cancer Res 1998 58: 3094–3100
Harding FA, Allison JP . CD28-B7 interactions allow the induction of CD8+ cytotoxic T lymphocytes in the absence of exogenous help J Exp Med 1993 177: 1791–1796
Nandi D, Gross JA, Allison JP . CD28-mediated costimulation is necessary for optimal proliferation of murine NK cells J Immunol 1994 152: 3361–3369
Chen PW, Ananthaswamy HM . Rejection of K1735 murine melanoma in syngeneic hosts requires expression of MHC class I antigens and either class II antigens or IL-2 J Immunol 1993 151: 244–255
Yang S, Darrow TL, Seigler HF . Generation of primary tumor specific CTL from autologous and HLA class I-matched allogeneic PBLs by B7 gene modified melanoma cells Cancer Res 1997 57: 1561–1568
Iezzi G, Protti MP, Rugarli C, Bellone M . B7.1 expression on tumor cells circumvents the need of professional antigen presentation for in vitro propagation of cytotoxic T cell lines Cancer Res 1996 56: 11–15
Fenton R, Turcovski-Corrales SM, Taub DD . Induction ofmelanoma antigen-specific cytotoxic T lymphocytes in vitro bystimulation with B7-expressing human melanoma cell J lines J Immunother 1998 21: 95–108
Cayeux S et al. Influence of gene-modified (IL-1, IL-4, and B7) tumor cell vaccines on tumor antigen presentation J Immunol 1997 158: 2834–2841
Armstrong TD, Clements VK, Ostrand-Rosenberg S . MHC class II-transfected tumor cells directly present antigen to tumor-specific CD4+ T lymphocytes J Immunol 1998 160: 661–666
Maric M et al. Maturation of cytotoxic T lymphocytes against a B7-transfected nonmetastatic tumor: a critical role of costimulation by B7 on both tumor and host antigen-presenting cells Cancer Res 1998 58: 3376–3384
Huang AYC et al. Role of bone marrow-derived cells in presenting MHC class I-restricted tumor antigens Science 1994 264: 961–965
Freeman GJ et al. Structure, expression, and T cell costimulatory activity of the murine homologue of the human b lymphocyte activation antigen B7 J Exp Med 1991 174: 625–631
Lewis JA . Biological assays for interferons. In: Clemens MJ, Morris AG, Gearing AJH (eds). Lymphokines and Interferons: A Practical Approach IRL Press: Washington DC, 1987, p73
Widmer MB, Voice RF, Conlon PJ, Grabstein KH . Precursor frequency and lytic specificity of interleukin 2 and interleukin 4-responsive murine lymphocytes Transplantation 1990 49: 743–748
Taswell C . Limiting dilution assays for the determination of immunocompetent cell frequencies J Immunol 1981 126: 1614–1619
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yang, S., Vervaert, C., Seigler, H. et al. Tumor cells cotransduced with B7.1 and γ-IFN induce effective rejection of established parental tumor. Gene Ther 6, 253–262 (1999). https://doi.org/10.1038/sj.gt.3300820
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3300820
Keywords
This article is cited by
-
Effective immunotherapy of weakly immunogenic solid tumours using a combined immunogene therapy and regulatory T-cell inactivation
Cancer Gene Therapy (2010)
-
Local gene therapy of solid tumors with GM-CSF and B7-1 eradicates both treated and distal tumors
Cancer Gene Therapy (2006)
-
High levels of Fas ligand and MHC class II in the absence of CD80 or CD86 expression and a decreased CD4+ T cell Infiltration, enables murine skin tumours to progress
Cancer Immunology, Immunotherapy (2003)