Abstract
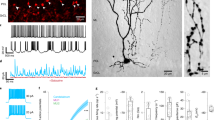
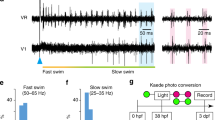
Glycine and γ-aminobutyric acid (GABA) are inhibitory transmitters of major importance1,2. Whereas neurones using GABA as the transmitter can be visualized by immunocytochemical methods for glutamate decarboxylase (GAD)3,4 or GABA5–8, no comparable techniques have been available for the selective visualization of glycinergic neurones. We have now produced polyclonal antibodies which specifically recognize glycine in glutaral-dehyde-fixed tissue. We used these antibodies to investigate the distribution of glycine in the simple central nervous system (CNS) of the Xenopus embryo, which contains an anatomically and physiologically defined class of reciprocal inhibitory interneurones, the commissural interneurones9–11. These interneurones have an important role in the generation of the swimming motor pattern and are thought to be glycinergic11–13. The glycine antibodies specifically stain these interneurones, revealing their distribution and number in the embryo CNS. This is the first demonstration of the selective localization of glycine-like immunoreactivity in a putative glycinergic class of neurone that has been characterized physiologically, pharmacologically and anatomically.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Curtis, D. R. & Johnston, G. A. R. Ergebn. Physiol. 69, 97–188 (1974).
Krnjević, K. Physiol Rev. 54, 418–540 (1974).
Saito, K. et al. Proc. natn. Acad. Sci. U.S.A. 71, 269–273 (1974).
Mugnaini, E. & Oertel, W. Handbook of Chemical Neuroanatomy Vol. 4 (eds Björklund, A. & Hökfelt, T.) (Elsevier, Amsterdam, 1985).
Storm-Mathisen, J. et al. Nature 301, 517–520 (1983).
Ottersen, O. P. & Storm-Mathisen, J. J. comp. Neurol. 229, 374–392 (1984).
Seguela, P., Geffard, M., Buijs, R. M. & Le Moal, M. Proc. natn. Acad. Sci U.S.A. 81, 3888–3892 (1984).
Hodgson, A., Penke, B., Erdei, A., Chubb, I.W. & Somogyi, P. J. Histochem. Cytochem. 33, 229–239 (1985).
Roberts, A. & Clarke, J. D. W. Phil Trans. R. Soc. B296, 195–212 (1982).
Soffe, S. R., Clarke, J. D. W. & Roberts, A. J. Neurophysiol 51, 1256–1267 (1984).
Dale, N. J. Physiol, Lond. 363, 61–70 (1985).
Roberts, A., Soffe, S. R. & Dale, N. in Neurobiology of Vertebrate Locomotion (eds Grillner, S. et al.) (Macmillan, London, 1986).
Soffe, S. R. J. Physiol, Lond. 382, (1987).
Werman, R., Davidoff, R. A. & Aprison, M. H. Nature 214, 681–683 (1967).
Curtis, D. R., Hösli, L. & Johnston, G. A. R. Expl Brain Res. 6, 1–18 (1968).
Davidoff, R. A., Graham, L. T. Jr., Shank, R. P., Werman, R. & Aprison, M. H. J. Neurochem. 14, 1025–1031 (1967).
Homma, S., Suzuki, T., Murayama, S. & Otsuka, M. J. Neurochem. 32, 691–698 (1979).
Hökfelt, T. & Ljungdahl, Å. Brain Res. 32, 189–194 (1971).
Ljungdahl, Å. & Hökfelt, T. Brain Res. 62, 587–595 (1973).
Matus, A. I. & Dennison, M. E. Brain Res. 32, 195–197 (1971).
Price, D. L., Stocks, A., Griffin, J. W., Young, A. & Peck, K. J. Cell Biol. 68, 389–395 (1976).
Pourcho, R. G. & Goebel, D. J. Brain Res. 348, 339–342 (1985).
Nieuwkoop, P. D. & Faber, J. Normal Tables of Xenopus laevis (Daudin) (North-Holland, Amsterdam, 1956).
Soffe, S. R. & Roberts, A. J. Neurophvsiol. 48, 1279–1288 (1982)
Dale, N., Roberts, A., Ottersen, O. P. & Storm-Mathisen, J. (in preparation).
Madsen, S., Ottersen, O. P. & Storm-Mathisen, J. Neurosci Lett. 60, 255–260 (1985).
Porath, J., Aspberg, K., Drevin, H. & Axén, R. J. Chromatogr. 86, 53–56 (1973).
Hardy, P. M., Nicholls, A. C. & Rydon, H. N. J.C.S. Perkin Trans. 9, 958–962 (1976).
Campistron, G., Buijs, R. M. & Geffard, M. Brain Res. 376, 400–405 (1986).
Ottersen, O. P., Davanger, S. & Storm-Mathisen, J. expl. Brain Res. (in the press).
Ottersen, O. P., Storm-Mathisen, J., Madsen, S., Skumlien, S. & StrøMathisen, J. med. Biol. 64, 147–158 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dale, N., Ottersen, O., Roberts, A. et al. Inhibitory neurones of a motor pattern generator in Xenopus revealed by antibodies to glycine. Nature 324, 255–257 (1986). https://doi.org/10.1038/324255a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/324255a0
This article is cited by
-
Immunogold cytochemistry in neuroscience
Nature Neuroscience (2013)
-
Immunogold quantification of amino acids and proteins in complex subcellular compartments
Nature Protocols (2008)
-
Roles for inhibition: studies on networks controlling swimming in young frog tadpoles
Journal of Comparative Physiology A (2008)
-
Increased expression of phosphate-activated glutaminase in hippocampal neurons in human mesial temporal lobe epilepsy
Acta Neuropathologica (2007)
-
The neuronal targets for GABAergic reticulospinal inhibition that stops swimming in hatchling frog tadpoles
Journal of Comparative Physiology A (2003)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.