Abstract
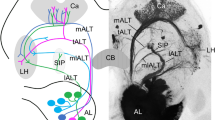
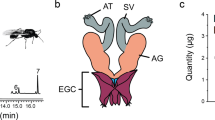
When tobacco hornworm moths (Manduca sexta) are tested in a wind tunnel with a source of female pheromones upwind, males but not normal females show pheromone-modulated anemotaxis and a characteristic mate-seeking behavioural sequence1–7. These behaviours are produced by stimulation of sensory neurones found only in male antennae8–10. These neurones project axons only to dendrites of pheromone-specific interneurones11,12 in the macroglomerular complex, a region of neuropil in the antennal lobe characteristic of males but not present in normal females13–15. Some interneurones in the antennal lobes of female moths that have received grafts of male antennae (gynandromorphs) respond postsynaptically to stimulation with bombykal, a major component of the pheromone16. They branch into a region resembling the macroglomerular complex16, like their counterparts in normal males. We show here that gynandromorphic females respond to pheromonal stimulation with anemotaxis. We also find that normal females display a similar sequence in response to the odour of their egg-laying site, the tobacco plant. It is likely that a common motor path is used either by pheromone-specific interneurones in the antennal lobes of males or by tobacco-specific interneurones in females. We assume that the interneurones in gynandromorphic females that branch into the macroglomerular complex induced by a grafted male antenna can activate this pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kennedy, J. S. & Marsh, D. Science 184, 999–1001 (1974).
Kennedy, J. S., Ludlow, A. R. & Sanders, C. J. Nature 288, 475–477 (1980).
Kennedy, J. S., Ludlow, A. R. & Sanders, C. J. Physiol. Ent. 6, 395–412 (1981).
Marsh, D., Kennedy, J. S. & Ludlow, A. R. Physiol. Ent. 6, 225 (1981).
Teal, P. E. A., McLaughlin, J. R. & Tumlinson, J. H. Ann. ent. Soc. Am. 74, 324–330 (1981).
Kuenen, L. P. S. & Baker, T. C. Physiol. Ent. 8, 277–289 (1983).
David, C. T., Kennedy, J. S. & Ludlow, A. R. Nature 303, 804–806 (1983).
Sanes, J. R. & Hildebrand, J. G. Devl Biol. 51, 282–299 (1976).
Kaissling, K.-E. in Chemical Ecology: Odour Communication in Animals (ed. Ritter, F. J.) 43–56 (Elsevier, Amsterdam, 1979).
Kaissling, K.-E. & Thorson, J. in Receptors for Neurotransmitters, Hormones, and Pheromones in Insects (eds Sattelle, D. B., Hall, L. M. & Hildebrand, J. G.) 261–282 (Elsevier, Amsterdam, 1980).
Schneiderman, A. M. thesis, Harvard Univ. (1984).
Christensen, T. A. & Hildebrand, J. G. Soc. Neurosci. Abstr. 10, 862, (1984).
Matsumoto, S. G. & Hildebrand, J. G. Proc. R. Soc. B213, 249–277 (1981).
Boeckh, J., Boeckh, V. & Kühn, A. Olfaction Taste, Paris 6, 315–321 (1977).
Olberg, R. M. Physiol. Ent. 8, 419–428 (1983).
Schneiderman, A. M., Matsumoto, S. M. & Hildebrand, J. G. Nature 298, 844–846 (1982).
Hildebrand, J. G. in Model Neural Networks and Behavior (ed. Selverston, A. I.) 129–148 (Plenum, New York, 1985).
Tolbert, L. P. & Hildebrand, J. G. Proc. R. Soc. B213, 279–301 (1981).
Nordlander, R. H. & Edwards, J. S. Wilhelm Roux Arch. dev. Biol. 162, 197–217 (1969).
Gregory, G. E. in Neuroanatomical Techniques: Insect Central Nervous System (eds Strausfeld, N. J. & Miller, T. A.) 75–95 (Springer, New York, 1980).
Hildebrand, J. G., Hall, L. M. & Osmond, B. C. Proc. natn. Acad. Sci. U.S.A. 76, 499–503 (1979).
Burrows, M., Boeckh, J. & Esslen, J. J. comp. Physiol 145, 447–457 (1982).
Ernst, K. D. & Boeckh, J. Cell Tissue Res. 229, 1–22 (1983).
Olberg, R. M. J. comp. Physiol. 152, 297–307 (1983).
Hildebrand, J. G. & Montague, R. A. in Mechanisms in Insect Olfaction (eds Payne, T., Birch, M. & Kennedy, C. E. J.) 278–285 (Oxford University Press, London, 1986).
Light, D. M. in Mechanisms in Insect Olfaction (eds Payne, T., Birch, M. & Kennedy, C. E. J.) 287–301 (Oxford University Press, London, 1986).
Marsh, D., Kennedy, J. S. & Ludlow, A. R. Physiol. Ent. 3, 221–240 (1978).
Miller, J. R. & Roelofs, W. L. J. chem. Ecol. 4, 187–198 (1978).
Baker, T. C. & Linn, C. E. Jr in Techniques in Pheromone Research (eds Hummel, H. E. & Miller, T. A.) 75–110 (Springer, New York, 1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schneiderman, A., Hildebrand, J., Brennan, M. et al. Trans-sexually grafted antennae alter pheromone-directed behaviour in a moth. Nature 323, 801–803 (1986). https://doi.org/10.1038/323801a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/323801a0
This article is cited by
-
Neural circuit evolved to process pheromone differently in two species of fruit fly
Nature (2018)
-
Neurotransmitter Mechanisms of Context-Dependent Behavior
Neuroscience and Behavioral Physiology (2014)
-
The joy of sex pheromones
EMBO reports (2013)
-
Embryonic assembly of a central pattern generator without sensory input
Nature (2002)
-
Moth flights of fancy
Nature (1994)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.