Abstract
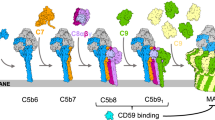
Cytolysis mediated by complement or cytolytic lymphocytes results in the formation of morphology similar lesions in the target membrane1–6. These lesions, formed by the polymerization of C9 or perforin respectively, contribute the major killing action by causing osmotic lysis of the target cell. Following the suggestion of Mayer7 that the mechanisms of humoral and cell-mediated cytotoxicity might be related, studies into the morphology of the membrane lesions formed8–13, and the proteins responsible for causing the lesions9,14,15, have shown several similarities. While the lesion caused by natural and T-killer cells is a little larger than that caused by complement, its overall shape is similar and in both cases the cylindrical pore is formed by polymerization of a monomeric subunit, C9 (relative molecular mass, Mr = 71,000) for complement9, and perforin (Mr = 66,000) for cell-mediated cytotoxicity14,15. C9 has an absolute requirement for a receptor in the target membrane formed by the earlier membrane attack complex components, C5b, C6, C7 and C8 (ref. 8). For perforin, polymerization in a target membrane requires no receptor, specificity being derived from the specific recognition between killer and target cell. Both proteins can be made to polymerize in vitro by the addition of divalent cations (Zn2+ for C9 (ref. 16) and Ca2+ for perforin10,12,13) and the resultant complexes closely resemble their physiological counterparts. Antibodies raised against lymphocyte-killed targets have also been shown to cross-react with complement proteins17,18,33, but the antigenically related proteins were not determined in these studies. We show here using purified proteins that perforin, C9 and complexes involving C7 and C8 share a common antigenic determinant which is probably involved in polymerization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bhakdi, S. & Tranum-Jensen, J. Biochim. biophys. Acta 737, 343–372 (1983).
Lachmann, P. J. & Thompson, R. A. J. exp. Med. 131, 643–652 (1970).
Müller-Eberhard, H. J. A. Rev. Biochem. 44, 697–723 (1975).
Lachmann, P. J. Nature 305, 473 (1983).
Podack, E. R. Immun. Today 6, 21–27 (1985).
Dourmashkin, R. R., Deteix, P., Simon, C. B. & Henkart, P. Clin. exp. Immun. 42, 554–559 (1980).
Mayer, M. M. J. Immun. 119, 1195–1202 (1977).
Tschopp, J., Müller-Eberhard, H. J. & Podack, E. R. Nature 298, 534–538 (1982).
Podack, E. R. & Tschopp, J. Proc. natn. Acad. Sci. U.S.A 79, 574–578 (1982).
Henkart, P. A., Millard, P. J., Reynolds, C. W. & Henkart, M. P. J. exp. Med. 160, 75–91 (1984).
Dennert, G. & Podack, E. R. J. exp. Med. 157, 1483–1495 (1983).
Masson, D., Corthésy, P., Nabholz, M. & Tschopp, J. EMBO J. 4, 2533–2538 (1985).
Podack, E. R. & Königsberg, P. J. J. exp. Med. 160, 695–710 (1984).
Masson, D. & Tschopp, J. J. biol. Chem. 260, 9096–9072 (1985).
Podack, E. R., Young, J. D. & Cohn, Z. A. Proc. natn. Acad. Sci. U.S.A. 82, 8629–8633 (1985).
Tschopp, J. J. biol. Chem. 259, 10569–10573 (1984).
Sundsmo, J. S. & Müller-Eberhard, H. J. J. Immun. 122, 2371–2378 (1975).
Ward, R. H. R. & Lachmann, P. J. Immunology 56, 179–188 (1985).
Stanley, K. K., Kocher, H. P., Luzio, J. P., Jackson, P. & Tschopp, J. EMBO J. 4, 375–382 (1985).
DiScipio, R. G. et al. Proc. natn. Acad. Sci. U.S.A. 81, 7298–7302 (1984).
Stanley, K. K., Page, M., Campbell, A. K. & Luzio, J. P. Molec. Immun. (in the press).
Tschopp, J. & Mollnes, T. E. Proc. natn. Acad. Sci. U.S.A. 83, 4223–4227 (1986).
George, D. G., Barker, W. C. & Hunt, L. T. Nucleic Acids Res. 14, 11–15 (1986).
Südhof, T. C., Goldstein, J. L., Brown, M. S. & Russel, D. W. Science 228, 815–822 (1985).
Schneider, W. J., Beisiegel, U., Goldstein, J. L. & Brown, M. S. J. biol. Chem. 257, 2664–2673 (1982).
Goldstein, J. L., Basu, S. K., Brunschede, G. Y. & Brown, M. S. Cell 7, 85–95 (1976).
Lämmli, U. K. Nature 227, 680–685 (1970).
Towbin, H., Staehelin, T. & Gordon, J. Proc. natn. Acad. Sci. U.S.A. 76, 4350–4358 (1979).
Podack, E. R. & Tschopp, J. Molec. Immun. 21, 589–603 (1984).
Atherton, E., Gait, M. J., Sheppard, R. C. & Williams, B. J. Bioorg. Chem. 8, 351–370 (1979).
Bassiri, R. M., Dvorak, J. & Utiger, R. D. in Methods of Hormone Radioimmunoassay (eds Jaffe, B. M. & Behrman, H. R.) 46–47 (Academic, New York, 1979).
Cosman, D. et al. Nature 312, 768–771 (1984).
Zalman et al. Bioscience rep. 5, 1093–1095 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tschopp, J., Masson, D. & Stanley, K. Structural/functional similarity between proteins involved in complement- and cytotoxic T-lymphocyte-mediated cytolysis. Nature 322, 831–834 (1986). https://doi.org/10.1038/322831a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/322831a0
This article is cited by
-
Beyond pore formation: reorganization of the plasma membrane induced by pore-forming proteins
Cellular and Molecular Life Sciences (2021)
-
Immune evolution from preneoplasia to invasive lung adenocarcinomas and underlying molecular features
Nature Communications (2021)
-
Low-grade inflammation as a key mediator of the pathogenesis of osteoarthritis
Nature Reviews Rheumatology (2016)
-
Perturbation of gut bacteria induces a coordinated cellular immune response in the purple sea urchin larva
Immunology & Cell Biology (2016)
-
Coexpression of human perforin improves yeast-mediated delivery of DNA and mRNA to mammalian antigen-presenting cells
Gene Therapy (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.