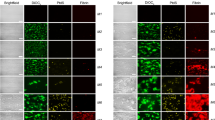
Abstract
Platelets activated by α-thrombin change shape from disks to spheres and aggregate. During this sequence of activation, platelets develop a membrane-bound lectin activity1,2 which is expressed after the secretion of intracellular granule contents3. This lectin activity seems to be important in mediating platelet aggregation by binding to a specific receptor on other platelets. Gartner et al.4 have recently shown that fibrinogen is the receptor for the endogenous lectin secreted by activated platelets. Thrombospondin5 (also called glycoprotein G6 and thrombin-sensitive protein7,8) is an α-granule constituent which has a molecular weight (MW) of 450,000 and is a disulphide-linked trimer of 160,000 MW subunits5,9,10. Secreted thrombospondin5–14 binds to platelet membranes in a calcium-dependent manner12. We show here that thrombospondin is the endogenous lectin of human platelets.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gartner, T. K., Williams, D. C. & Phillips, D. R. Biochem. biophys. Res. Commun. 79, 592–599 (1977).
Gartner, T. K., Williams, D. C., Minion, F. C. & Phillips, D. R. Science 200, 1281–1283 (1978).
Gartner, T. K., Phillips, D. R. & Williams, D. C. FEBS Lett. 113, 196–200 (1980).
Gartner, T. K., Gerrard, J. M., White, J. G. & Williams, D. C. Nature 289, 688–690 (1981).
Lawler, J. W., Slayter, H. S. & Coligan, J. E. J. biol. Chem. 253, 8609–8616 (1978).
George, J. N. J. Lab. clin. Med. 92, 430–446 (1978).
Baenziger, N. D., Brodie, G. N. & Majerus, P. W. Proc. natn. Acad. Sci. U.S.A. 68, 240–243 (1971).
Baenziger, N. L., Brodie, G. N. & Majerus, P. W. J. biol. Chem. 247, 2723–2731 (1972).
Hagen, I. Biochim. biophys. Acta 392, 242–254 (1975).
Phillips, D. R. & Agin, P. P. J. biol. Chem. 252, 2121–2126 (1977).
Lawler, J. W., Chao, F. C. & Fang, P. H. Thromb. Haemostasis 37, 355–357 (1977).
Phillips, D. R., Jennings, L. K. & Prasanna, H. R. J. biol. Chem. 255, 11629–11632 (1980).
George, J. N., Lyons, R. M. & Morgan, R. K. J. clin. Invest. 66, 1–9 (1980).
Hagen, I., Olsen, T. & Solum, N. O. Biochim. biophys. Acta 455, 214–225 (1976).
Phillips, D. R. & Gartner, T. K. in Receptors and Recognition Ser. B, Vol. 6 (ed. Beachey, E. H.) 399–438 (Chapman & Hall, London, 1980).
Gerrard, J. M. et al. J. clin. Invest. 66, 102–109 (1980).
Hawiger, J., Fujimoto, T. & Ohara, S. Fedn Proc. 40, 833 (1981).
Tschopp, T., Weiss, H. J. & Baumgartner, H. J. Lab. clin. Med. 83, 296–300 (1974).
Weiss, H. J., Baumgartner, H. R., Tschopp, T. B., Turitto, V. T. & Cohen, D. Blood 51, 267–279 (1978).
Miletich, J. P., Jackson, C. M. & Majerus, P. W. J. biol. Chem. 253, 6908–6916 (1978).
Tracy, P. B., Peterson, J. M., Nesheim, M. E., McDuffie, F. C. & Mann, K. G. J. biol. Chem. 254, 10354–10361 (1979).
Kane, W. H., Lindhout, M. J., Jackson, C. M. & Majerus, P. W. J. biol. Chem. 255, 1170–1174(1980).
Mustard, J. F., Packham, M. A., Kinlough-Rathbone, R. L., Perry, D. W. & Regoeczi, E. Blood 52, 453–466 (1978).
Marguerie, G. A., Plow, E. F. & Edgington, T. S. J. biol. Chem. 254, 5357–5363 (1979).
Bennett, J. S. & Vilaire, G. J. clin. Invest. 65, 1393–1401 (1979).
Plow, E. F. & Ginsberg, M. H. J. biol. Chem. 256, 9477–9482 (1981).
Jaffe, E. A., Hoyer, L. W. & Nachman, R. L. J. clin. Invest. 52, 2757–2764 (1973).
Jaffe, E. A., Hoyer, L. W. & Nachman, R. L. Proc. natn. Acad. Sci. U.S.A. 71, 1906–1909 (1974).
Jaffe, E. A. & Mosher, D. F. J. exp. Med. 147, 1779–1791 (1978).
Doyle, M. J., Mosher, D. F. & Jaffe, E. A. J. Cell Biol. 87, 306a (1980).
Mosher, D. F., Doyle, M. J. & Jaffe, E. A. J. Cell Biol. (in the press).
Wolfenstein-Todel, C. & Mosesson, M. W. Proc. natn. Acad. Sci. U.S.A. 77, 5069–5073 (1980).
Jaffe, E. A. & Nachman, R. L. J. clin. Invest. 56, 698–702 (1975).
Deutsch, D. G. & Mertz, E. T. Science 170, 1095–1096 (1970).
Engvall, E. & Ruoslahti, E. Int. J. Cancer 20, 1–5 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jaffe, E., Leung, L., Nachman, R. et al. Thrombospondin is the endogenous lectin of human platelets. Nature 295, 246–248 (1982). https://doi.org/10.1038/295246a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/295246a0
This article is cited by
-
Thrombospondin as a mediator of cancer cell adhesion in metastasis
Cancer and Metastasis Reviews (1992)
-
Thrombospondin expression in traumatized skeletal muscle
Cell and Tissue Research (1990)
-
Immunoelectron-microscopic studies of human platelet thrombospondin, Von Willebrand factor, and fibrinogen redistribution during clot formation
The Histochemical Journal (1990)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.