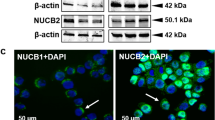
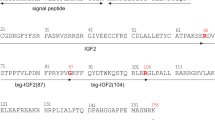
Abstract
The pituitary ACTH/MSH cells are virtual storehouses for biologically important peptides. In addition to corticotropins, melanotropins, opioid peptides and lipolytic peptides these cells contain large amounts of at least three different polypeptides (or small proteins) having tryptophan in the amino-terminal position (Trp-peptides)1–5; their approximate molecular weight (MW) has been given as 14,000 (ref. 4). Nothing is known of their chemical properties and physiological significance. Experimental data indicate that the Trp-peptides in the ACTH cells are stored in the secretory granules3 to be released jointly with ACTH and other secretory products, such as β-endorphin and/or β-lipotropin (β-LPH)6. ACTH and β-LPH are formed from a large common precursor protein where they constitute the carboxy-terminal portion7–14. Together the two peptides account for one-third to one-half of the molecular weight of the precursor and there has been considerable interest in the primary structure and possible physiological significance of the remaining amino-terminal portion15. The fact that the Trp-peptides occur in conspicuously high concentrations in the secretory granules of the ACTH/MSH cells prompted an investigation into their relation to the ACTH-β-LPH precursor protein. We have now isolated a glycopeptide with an apparent MW of ∼11,000 residing in the secretory granules of the ACTH/MSH cells from pig pituitary. The amino acid sequence of its amino-terminal part was found to be identical with the amino-terminal end of the bovine corticotropin-β-LPH precursor protein16. We propose that the isolated new fragment is identical with the amino-terminal portion of the precursor that remains after ACTH and β-lipotropin have been split off.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Håkanson, R., Larsson, L.-I., Nobin, A. & Sundler, F. J. Histochem. Cytochem. 20, 908–916 (1972).
Håkanson, R. et al. Cell Tissue Res. 150, 281–290 (1974).
Håkanson, R., Sundler, F., Larsson, L.-I., Ekman, R. & Sjöberg, N.-O. J. Histochem. Cytochem. 23, 65–74 (1975).
Håkanson, R., Ekman, R. & Sundler, F. Acta pharmac. toxicol. (Kbh) Suppl. 41, 50 (1977).
Partanen, S. Histochemistry 56, 147–154 (1978).
Håkanson, R. & Sundler, F. Cell Tissue Res. 183, 419–421 (1977).
Nakanishi, S. et al. Proc. natn. Acad. Sci. U.S.A. 73, 4319–4323 (1976).
Nakanishi, S., Inoue, A., Taii, S. & Numa, S. FEBS Lett. 84, 105–109 (1977).
Taii, S., Nakanishi, S. & Numa, S. Eur. J. Biochem. 93, 205–212 (1979).
Kita, T., Inoue, A., Nakanishi, S. & Numa, S. Eur. J. Biochem. 93, 213–220 (1979).
Mains, R. E. & Eipper, B. A. J. biol. Chem. 251, 4115–4120 (1976).
Mains, R. E., Eipper, B. A. & Ling, N. Proc. natn. Acad. Sci. U.S.A. 74, 3014–3018 (1977).
Eipper, B. A. & Mains, R. E. J. biol. Chem. 253, 5732–5744 (1978).
Roberts, J. L. & Herbert, E. Proc. natn. Acad. Sci. U.S.A. 74, 4826–4830, 5300–5304 (1977).
Hughes, J. Nature 278, 394–395 (1979).
Nakanishi, S. et al. Nature 278, 423–427 (1979).
Austen, B. M. & Smyth, D. G. Endocrinology 79, 19 (abs) (1979).
Eipper, B. A., Mains, R. E. & Guenzi, D. J. biol. Chem. 251, 4121–4126 (1976).
Björklund, A., Meurling, P., Nilsson, G. & Nobin, A. J. Endocr. 53, 161–169 (1972).
Schwyzer, R. Ann. N.Y. Acad. Sci. 297, 3–26 (1977).
Schwandt, P., Richter, W. & Wilkening, J. FEBS Lett. 100, 360–362 (1979).
de Moor, P., Osinski, P., Deckx, R. & Steeno, O. Clin. chim. Acta 7, 475–480 (1962).
Miller, R. J., Chang, K. J., Leighton, J. & Cuatrecasas, P. Life Sci. 22, 379–387 (1978).
Håkanson, R. & Sundler, F. Biochem. Pharmac. 20, 3223–3225 (1971).
Lowry, O. H., Rosenbrough, N. J., Farr, A. L. & Randall, R. J. J. biol. Chem. 193, 265–275 (1951).
Spackman, D. M., Stein, W. H. & Moore, S. Analyt. Chem. 30, 1190–1206 (1958).
Moore, S. J. biol. Chem. 238, 235–237 (1963).
Penke, B., Ferenczi, R. & Kovaćs, K. Analyt. Biochem. 60, 45–50 (1974).
Edelhoch, H. Biochemistry 6, 1948–1954 (1967).
King, J. & Laemmli, U.K. J. molec. Biol. 62, 465–477 (1971).
Sternberger, L. A. Immunocytochemistry, 129 (Prentice-Hall, Englewood Cliffs, 1974).
Edman, P. & Begg, G. Eur. J. Biochem. 1, 80–91 (1967).
Tarr, G.E., Beecher, J.F., Bell, M. & McKean, D.J. Analyt. Biochem. 84, 622–627 (1978).
Klapper, D. G., Wilde, C. E. & Capra, J. D. Analyt. Biochem. 85, 126–131 (1978).
Jeppsson, J.-O. & Sjöquist, J. Analyt. Biochem. 18, 264–269 (1967).
Keutmann, H. T., Eipper, B. A. & Mains, R. E. J. biol. Chem. 254, 9204–9208 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Håkanson, R., Ekman, R., Sundler, F. et al. A novel fragment of the corticotropin-β-lipotropin precursor. Nature 283, 789–792 (1980). https://doi.org/10.1038/283789a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/283789a0
This article is cited by
-
The effect of reserpine on the pars intermedia of the rat pituitary
Cell and Tissue Research (1985)
-
Pro-opiocortin: The mutiple adrenal hormone precursor
Bioscience Reports (1984)
-
Evidence for the presence of pro-?-melanotropin, the NH2-terminal fragment of the corticotropin-?-lipotropin precursor, in corticotropin-producing tumours
Virchows Archiv A Pathological Anatomy and Histology (1981)
-
A novel human pituitary peptide containing the γ-MSH sequence
Nature (1980)
-
Pituitary immunoreactive γ-melanotropins are glycosylated oligopeptides
Nature (1980)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.