Abstract
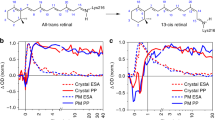
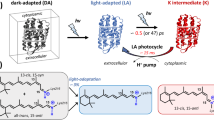
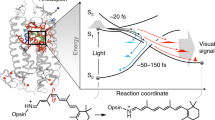
RHODOPSIN is the visual pigment in the disk membranes of vertebrate rod photoreceptors. It consists of a chromophore, 11-cis-retinal, covalently bound in the form of a protonated Schiff base to an ɛ-amino group of a lysine in its apoprotein opsin. The primary photochemical event in visual excitation involves the formation of a species known as bathorhodopsin1 which has been detected at room temperature2 and at 4 K (ref. 3). The rhodopsin–bathorhodopsin transformation has been thought to involve a photochemical 11-cis → all-trans isomerisation; however, this conclusion has recently been questioned2–5 primarily because bathorhodopsin is formed within a few picoseconds at room temperature2 and in 36 ps at 4 K (ref. 3), and because of the large isotope effect involved in its formation3. In this letter, we show on the basis of resonance Raman experiments that isorhodopsin (the artificial pigment with 9-cis-retinal as its chromophore) is formed photochemically from rhodopsin at liquid helium temperatures. All models for the primary event other than isomerisation about the 11–12 double bond are found to be inconsistent with this result. We present a specific model (see also ref. 6) for the formation of bathorhodopsin which accounts for all the available data.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Yoshizawa, T. & Wald, G. Nature 197, 1279–1286 (1963).
Busch, G., Applebaum, M., Lamola, A. & Rentzepis, P. Proc. natn. Acad. Sci. U. S. A. 69, 2802–2806 (1972).
Peters, K., Applebury, M. L. & Rentzepis, P. M. Proc. natn. Acad. Sci. U. S. A. 74, 3119–3123 (1977).
van der Meer, K., Mulder, J. J. C. & Lugtenberg, J. Photochem. Photobiol. 24, 363–367 (1976).
Huppert, D., Rentzepis, P. M. & Kliger, D. Photochem. Photobiol. 25, 193–197 (1977).
Hurley, H. B., Ebrey, T. G., Honig, B. & Ottolenghi, M. Nature 270, 540–542 (1977).
Rosenfeld, T., Honig, B., Ottolenghi, M., Hurley, J. & Ebrey, T. G. Pure Appl. Chem. 49, 341–351 (1977).
Hubbard, R. & Kropf, A. Proc. natn. Acad. Sci. U. S. A. 44, 130–139 (1958).
Aston, J. C., Szasz, G., Wooley, H. W. & Brickwedde, F. G. J. chem. Phys. 14, 67–77 (1946).
Yoshizawa, T. in Biochemistry and Physiology of Visual Pigments (ed. Langer, H.) 169–181 (Springer, Berlin, 1973).
Callender, R. H. & Honig, B. A. Rev. Biophys. Bioengng 6, 33–55 (1977).
Applebury, M. L., Zuckerman, D. M., Lumola, A. A. & Jovin, T. M. Biochemistry 13, 3448–3457 (1974).
Oseroff, A. R. & Callender, R. H. Biochemistry 13, 4243–4248 (1974).
Mathies, R., Freedman, T. B. & Stryer, L. J. molec. Biol. 109, 367–372 (1977).
Green, B. H., Monger, T. G., Alfano, R. R., Aton, B. & Callender, R. H. Nature 269, 179–180 (1977).
Honig, B., Greenberg, A. D., Dinur, U. & Ebrey, T. G. Biochemistry 15, 4593–4599 (1976).
Schaffer, A. M., Yamaoka, T. & Becker, R. S. Photochem. Photobiol. 21, 297–301 (1975).
Warshel, A. A. Rev. Biophys. Bioengng 6, 273–300 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ATON, B., CALLENDER, R. & HONIG, B. Photochemical cis–trans isomerisation of bovine rhodopsin at liquid helium temperatures. Nature 273, 784–786 (1978). https://doi.org/10.1038/273784a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/273784a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.