Abstract
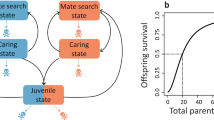
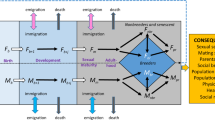
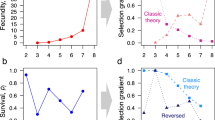
THE theory of R. A. Fisher1 for the effect of natural selection on the sex ratio predicts the population to be in evolutionary equilibrium when half of the parental reproductive resources are devoted to sons, and half to daughters. Provided that sons and daughters are of equal cost, this implies a sex ratio of ½ males at or near conception, and the result is independent of differential male and female survival after the period of parental care2–5. Most models demonstrating these results assume discrete generations, but some show that they also follow with overlapping generations6–8. These latter models typically assume the population to be in stable age distribution. Here, we relax that assumption, and show that selection can favour genes which result in the temporary overproduction of one or the other sex, under certain general conditions. The conditions are; overlap in generations; different temporal changes in life history expectations for the two sexes; parental ability to vary the sex ratio in response to the life history changes. The results are of interest because stable age distributions are probably uncommon in nature, with fluctuations in survival more the norm.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fisher, R. A. The Genetical Theory of Natural Selection (Oxford University Press, 1929).
Shaw, R. F. & Mohler, J. D. Am. Nat. 87, 337–342 (1953)
Kolman, W. Am. Nat. 94, 373–377 (1960).
Bodmer, W. F. & Edwards, A. W. F. Ann. hum. Gen. 24, 239–244 (1960).
MacArthur, R. H in Theoretical and Mathematical Biology (eds Waterman T & Morowitz, H.) 388–397 (Blaisdell, Lexington, 1965).
Leigh, E. Am. Nat. 104, 205–210 (1970).
Charnov, E. L. Evolution 29, 366–368 (1975).
Leigh, E. G., Charnov, E. L. & Warner, R. R. Proc. natn. Acad. Sci. U.S.A. 73, 3565–3660 (1976).
Latham, R. M. J. Windl. Mant. Bull., Ottawa 11, 139–149 (1947).
Hamilton, J. B. Recent Prog. Horm. Res. 3, 257–322 (1948).
Flanders, S. E. Insectes Soc. 3, 325–334 (1956).
Clausen, C. P. J. N.Y. ent. Soc. 47, 1–9 (1939).
Flanders, S. E. Q. Rev. Biol. 21, 135–143 (1946).
Bouletrean, M. Ent. Exp. Appl. 19, 197–204 (1976).
Hoelscher, C. E. & Vinson, S. B. Ann. ent. Soc. Am. 64, 1373–1376 (1971).
Filipponi, A., Mosna, B. & Petrelli, G. Riv. Parasitt. 32, 193–218 (1972).
Filipponi, A. & Petrelli, G. Riv. Parasitt. 36, 295–308 (1975).
Evans, F. C. J. Mammal. 30, 351–363 (1949).
Petrusewicz, K. Acta theriol. 4, 103–137 (1960).
Krebs, C. J., Gaines, M., Keller, B., Meyers, J. & Tamarin, R. Science 179, 35–41 (1973).
Kalela, O. Ann. Zool. Fenn. 8, 452–455 (1971).
Canham, R. P. Can. J. Zool. 48, 809–811 (1970).
Fordham, R. A. Ecology 52, 138–146 (1971).
Nanmov, S. P., Gibet, L. A. & Shatlova, S. P. Zh. obshch. Biol. 30, 673–680 (1967).
Geodakian, V. A., Kosobutsky, V. I. & Bileva, D. S. Genetika 9, 154–163 (1967).
Correns, C. Hanb. Vererbursw. 2, 1–138 (1928).
Rychlewsk, J. & Kazimierez, Z. Acta biol. cracov. 18, 101–114 (1975).
Hertwig, R. Biol. Zbl. 32, 1–146 (1912).
Kuschakewitsch, S. Festschrift R. Hertwig 2, 61–224 (1910).
Huxley, J. S. J. Genet. 10, 265–276 (1920).
James, H. C. Proc. R. ent. Soc. Lond. 12, 92–98 (1937).
Mrsic, W. Arch. Mikr. Anat. 98, 129–209 (1923).
Hannah, A. Z. Ord. Abst. Vererbl. 86, 574–599 (1955).
Seiler, J. Retz, Arch. Zellforsch. 15, 249–268 (1920).
Volkmann-Rocco, B. Biol. Bull. 142, 520–529 (1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WERREN, J., CHARNOV, E. Facultative sex ratios and population dynamics. Nature 272, 349–350 (1978). https://doi.org/10.1038/272349a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/272349a0
This article is cited by
-
Transgenerational effects of thermal stress on reproductive physiology of fall armyworm, Spodoptera frugiperda
Journal of Pest Science (2023)
-
Endosymbionts moderate constrained sex allocation in a haplodiploid thrips species in a temperature-sensitive way
Heredity (2022)
-
Sex ratios deviate across killifish species without clear links to life history
Evolutionary Ecology (2020)
-
The global male-bias in sex ratio at birth is sustained by the sex ratio genotypes of replacement offspring
Genetica (2019)
-
Effect of short-term high-temperature exposure on the life history parameters of Ophraella communa
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.