Abstract
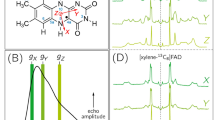
NUCLEAR magnetic resonance methods have provided a wealth of information on the conformational and ligand-binding properties of proteins1–3. In such studies, it is necessary to resolve resonances from the ligand or from individual residues of the protein. To achieve this, it is often necessary to resort to isotopic substitution methods such as selective deuteration4,5 or 13C enrichment6–8. An alternative approach is to introduce a fluorine atom (19F) into the system as an NMR probe, either by using fluorine-containing ligands9–11 or by labelling the protein itself. This can be done either by chemical modification12–15 or by the biosynthetic incorporation of selected fluorinated amino acids16–19. We have used this latter approach to prepare Lactobacillus casei dihydrofolate reductase containing either 3-fluoro-tyrosine or 6-fluorotryptophan residues; by measuring the 19F spectra at 94.1 MHz we have characterised the effects of ligand binding on individual tyrosine and tryptophan residues in the protein19. We now report through-space 19F–19F spin–spin coupling in 6-fluorotryptophan containing dihydrofolate reductase which demonstrates that two of the tryptophan residues are in close proximity in the folded structure of the protein.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Dwek, R. A. Nuclear Magnetic Resonance in Biochemistry Ch. 6 (Clarendon, Oxford, 1973).
Roberts, G. C. K. & Jardetzky, O. Adv. Protein Chem. 24, 447–545 (1970).
Wüthrich, K. NMR in Biological Research, Peptides and Proteins (Elsevier, Amsterdam 1976).
Markley, J. L., Putter, I. & Jardetzky, O. Science 161, 1249–1251 (1968).
Feeney, J. et al. Proc. R. Soc. Lond. B. 196, 267–290 (1977).
Hunkapiller, M. W., Smallcombe, S. H., Whitaker, D. R. & Richards, J. H. Biochemistry 12, 4732–4743 (1973).
Browne, D. T., Earl, E. M. & Otvos, J. D. Biochem. biophys. Res. Commun. 72, 398–403 (1976).
Feeney, J., in New Techniques in Biophysics and Cell Biology 2, Ch. 8 (eds Pain, R. H. & Smith, B. J.) (Wiley, New York, 1975).
Millet, F. & Raftery, M. A. Biochem. biophys. Res. Commun. 47, 625–632 (1972).
Dwek, R. A., Kent, P. W. & Xavier, A. V. Eur. J. Biochem. 23, 343–348 (1971).
Ashton, H., Capon, B. & Foster, R. L. Chem. Commun. 512–513 (1971).
Bode, J., Blumenstein, M. & Raftery, M. A. Biochemistry 14, 1153–1160 (1975).
Heustis, W. H. and Raftery, M. A. Biochemistry 14, 1886–1892 (1975).
Bendall, M. R. & Lowe, G. Eur. J. Biochem. 65, 493–502 (1976).
Critz, W. J. & Martinez-Carrion, M. Biochemistry 16, 1559–1564 (1977).
Hull, W. E., Sykes, B. D. Biochemistry 15, 1535–1546 (1976).
Coleman, J. E., Anderson, R. A., Ratcliffe, G. & Armitage, I. M. Biochemistry 15, 5419–5430 (1976).
Robertson, D. E., Kroon, P. A. & Ho, C. Biochemistry 16, 1443–1451 (1977).
Kimber, B. J. et al. Biochemistry 16, 3492–3500 (1977).
Campbell, I. D., Dobson, C. M., Williams, R. J. P. & Wright, P. E. FEBS Lett. 57, 96–99 (1975).
Ng, S. & Sederholm, H. J. Chem. Phys. 40, 2090–2094 (1964).
Petrakis, L., Sederholm, C. H. J. Chem. Phys. 35, 1243–1248 (1961).
Jonas, J. J. Chem. Phys. 47, 4884–4888 (1967).
Buckingham, A. D. & Cordle, J. E. J. Chem. Soc. Faraday Trans. II, 994–1004 (1974).
Hilton, J. & Sutcliffe, L. H. Prog. NMR Spectroscopy 10, 27–39 (1975).
Hilton, J. & Sutcliffe, L. H. Spectrochim Acta. 32A, 201–213 (1976).
Balaram, P., Bothner-By, A. A. & Dadok, J. J. Am. chem. Soc. 94, 4015–4017 (1972).
Bothner-By, A. A. & Gassend, R. Ann. N. Y. Acad. Sci. 222, 668–676 (1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KIMBER, B., FEENEY, J., ROBERTS, G. et al. Proximity of two tryptophan residues in dihydrofolate reductase determined by 19F NMR. Nature 271, 184–185 (1978). https://doi.org/10.1038/271184a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/271184a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.