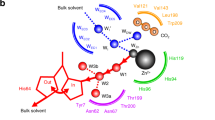
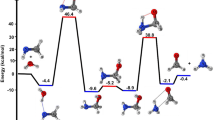
Abstract
THE hydration of carbon dioxide by Zn(II) carbonic anhydrase is thought1,2 to occur through a mechanism in which a hydroxyl group coordinated to the zinc atom has an important function. As a consequence, the pK of the conjugate acid coordinated water molecule has been the subject of some discussion2,3. In Zn(II) (ref. 4) and Co(II) (ref. 5) carbonic anhydrase, the water molecule bound to the metal ion, apparently has a pK in the range 6.9–7.9, which is two units lower than the pK range quoted6 for the first protolysis of [Zn(H2O)6]2+ and [Co(H2O)6]2+.(The pK values quoted for [M(H2O)6]2+ exhibit a considerable variance and have been determined under a variety of experimental conditions. When M = Co, Ni, Cu and Zn mean pK values of 9.8, 10.3, 7.8 and 9.5, respectively, are derived from the range of values quoted at zero ionic strength and at 298 K (pK values for all four metal ions in 1 M NaClO4 are not available).)
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lindskog, S., and Coleman, J. E., Proc. natn., Acad. Sci. U.S.A., 70, 2505 (1973).
Prince, R. H., and Woolley, P. R., Angew. Chem., Int. Ed. 11, 408 (1972).
Prince, R. H., and Woolley, P. R., Nature phys. Sci., 240, 117 (1972).
Bradbury, S. L., J. biol. Chem., 244, 2002, 2010 (1969).
Ward, R. L., and Fritz, K. J., Biochem. biophys. Res. Commun., 39, 707 (1970).
Sillen, L. G., and Martell, A. E., in Stability Constants of Metal Ion Complexes, Chemical Society Special Publication No 17., 1964., and supplement No.1, 1971.
Liljas, A., et al., Nature new Biol., 235, 131 (1972).
Beaumont, R. C., Inorg. Chem., 8, 1805 (1969).
Couldwell, M. C., House, D. A., and Powell, H. K. J., Aust. J. Chem., 26, 425 (1973).
Paoletti, P., Ciampolini, M., and Sacconi, L., J. chem. Soc., 3589 (1963).
Paoletti, P., and Ciampolini, M., Inorg. Chem., 6, 64 (1967).
Ciampolini, M., and Paoletti, P., Inorg. Chem., 6, 1261 (1967).
Orioli, P. L., Coordn. Chem. Rev., 6, 285 (1971).
Criotini, A., and Ponticelli, G., J. inorg. nucl. Chem., 35, 2691 (1973).
Jain, P. C., and Lingafelter, E. C., J. Am. chem. Soc., 89, 6131 (1967).
Wells, P. R., in Linear Free Energy Relationships (Academic Press, London, 1968).
Hepler, L. G., J. Am. chem. Soc., 85, 3089 (1963).
Pitzer, K. S., J. Am. chem. Soc., 59, 2365 (1937).
Frank, H. S., and Wen, W. Y., Discuss. Faraday Soc., 24, 133 (1957).
Klotz, I. M., Brookhaven Symp. quant. Biol., 13, 25 (1960).
Klotz, I. M., Science, 128, 815 (1958).
Mandel, J., and Linnig, F. J., Analyt. Chem., 29, 743 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
COATES, J., GENTLE, G. & LINCOLN, S. Influence of local hydrophobic environment on acid dissociation constants of coordinated water molecules. Nature 249, 773–775 (1974). https://doi.org/10.1038/249773a0
Received:
Issue Date:
DOI: https://doi.org/10.1038/249773a0
This article is cited by
-
Designing Heterogeneous Catalysts by Incorporating Enzyme-Like Functionalities into MOFs
Topics in Catalysis (2010)
-
Ab initio study of the HCO 3 ? /H2O exchange in the (NH3)3 ZnII(HCO 3 ? ) complex
Theoretica Chimica Acta (1995)
-
Models for metal ion function in carbonic anhydrase
Nature (1975)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.