Abstract
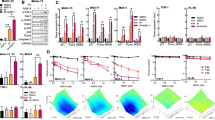
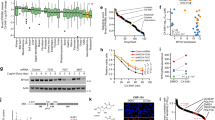
The use of topoisomerase inhibitors has been associated with the development of secondary malignancies, suggesting that these agents can induce DNA damage that may be persistent. We have investigated the effect of short exposures (>3 days) to low etoposide concentrations (LC-etoposide, 0.01–0.04 μM) on the ability of leukaemic cells to initiate apoptosis. Results showed that although LC-etoposide had no effect on cell growth characteristics, the pre-culture of cells with LC-etoposide conferred resistance to subsequent exposure to cytotoxic concentrations of etoposide (0.3 μM etoposide in HL60 on day 3: %V: 95.2 ± 1.6% vs 60.3 ± 12.1% in control cells with no pre-culture, and %A: 5.1 ± 0.2 vs 19.0 ± 0.7%; P < 0.001). This effect was still observed 4 weeks after the initial drug exposure. Associated with these observations was a three-fold increase in genetic instability and a reduction in induced bax protein levels. The anti-cytotoxic effect was also shown to be specific to topoisomerase II (topo II) inhibitors, as the pre-culture of cells with a low doxorubicin concentration also induced resistance, while low cisplatin concentrations did not. The persistence of these alterations in cellular processes following an initial exposure to topo II inhibitors suggests a DNA-based mechanism, and highlights the existence of drug/target interactions even at very low drug concentrations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Clark PI, Slevin ML . The clinical pharmacology of etoposide and teniposide Clin Pharmacokinet 1987 v 12: 223–252
Slevin ML, Clark PI, Joel SP, Malik S, Osborne RJ, Gregory WM, Lowe DG, Reznek RH, Wrigley PF . A randomised trial to evaluate the effect of schedule on the activity of etoposide in small cell lung cancer J Clin Oncol 1989 7: 1333–1340
Pui CH, Ribeiro RC, Hancock ML, Rivera GK, Evans WE, Raimondi SC, Head DR, Behm FG, Mahmoud MH, Sandlund JT . Acute myeloid leukaemia in children treated with epipodophyllotoxins for acute lymphocytic leukaemia N Engl J Med 1991 325: 1682–1687
Winick NJ, McKenna RW, Shuster JJ, Schneider NR, Borowitz MJ, Bowman WP, Jacaruso D, Kamen BA, Buchanan GR . Secondary acute myeloid leukaemia in children with acute lymphoblastic leukaemia treated with etoposide J Clin Oncol 1993 11: 209–217
Smith MA, Rubinstein L, Ungerleider RS . Therapy-related acute myeloid leukaemia following treatment with epipodophyllotoxins: estimating the risks Med Pediatr Oncol 1994 23: 86–98
Smith MA, Rubinstein L, Anderson JR, Arthur D, Catalano PJ, Freidlin B, Heyn R, Khayat A, Krailo M, Land VJ, Miser J, Shuster J, Vena D . Secondary leukaemia or myelodysplatic syndrome after treatment with epipodophyllotoxins J Clin Oncol 1999 17: 569–577
Stine KC, Saylors RL, Sawyer JR, Becton DL . Secondary acute myelogenous leukaemia following safe exposure to etoposide J Clin Oncol 1997 15: 1583–1586
Wang JC . DNA topoisomerases Annu Rev Biochem 1985 54: 665–697
Zwelling LA . DNA topoisomerase II as a target of antineoplastic drug therapy Cancer Metastasis Rev 1985 4: 263–276
Berger NA, Chatterjee S, Schmotzer JA, Helms SR . Etoposide (VP-16–213)-induced gene alterations: potential contribution to cell death Proc Natl Acad Sci USA 1991 88: 8740–8743
Han YH, Austin MJ, Pommier Y, Povirk LF . Small deletions and insertion mutations induced by topoisomerase II inhibitor teniposide in CHO cells and comparison with sites of drug-stimulated DNA cleavage in vitro J Mol Biol 1993 220: 52–66
Liu WM, Bamford C, Slevin M, Joel SP . Effects of haemopoietic growth factors in combination with etoposide on sister chromatid exchange frequencies in peripheral blood mononuclear cells Cancer Chemother Pharmacol 1998 41: 343–346
Chatterjee S, Trivedi D, Petzold SJ, Berger NA . Mechanism of epipodophyllotoxin-induced cell death in poly(adenosine diphosphate-ribose) synthesis-deficient V79 Chinese hamster cell lines Cancer Res 1990 50: 2713–2718
Maraschin J, Dutrillaux, Aurias A . Chromosome aberrations induced by etoposide (VP-16) are not random Int J Cancer 1990 46: 808–812
Gromova II, Thomsen B, Razin SV . Different topoisomerase II antitumour drugs direct similar specific long-range fragmentation of an amplified c-MYC gene locus in living cells and in high-salt-extracted nuclei Proc Natl Acad Sci USA 1995 92: 102–106
Tkachuk DC, Kohler S, Cleary ML . Involvement of a homolog of Drosophila trithorax by 11q23 chromosomal translocations in acute leukaemias Cell 1992 71: 691–700
Ridge SA, Wiedemann LM . Chromosome 11q23 abnormalities in leukaemia Leuk Lymphoma 1994 14: 11–17
Joel SP . The clinical pharmacology of etoposide: an update Cancer Treat Rev 1996 22: 179–221
Liu WM, Lawrence AJ, Joel SP . The importance of drug scheduling and recovery phases in determining drug activity: improving etoposide efficacy in BCR-ABL-positive CML cells Eur J Cancer 2002 38: 842–850
Darzynkiewicz Z, Li X, Gong J . Assays of cell viability: discrimination of cells dying by apoptosis Methods Cell Biol 1994 41: 15–38
Mitelman F . ISCN An International System for Human Cytogenetic Nomenclature S Karger: Basel 1995
Klein I, Sarkadi B, Varadi A . An inventory of the human ABC proteins Biochim Biophys Acta 1999 1461: 237–262
Hamilton KO, Topp E, Makagiansar I, Siahaan T, Yazdanian M, Audus KL . Multidrug resistance-associated protein-1 functional activity in Calu-3 cells J Pharmacol Exp Ther 2001 298: 1199–1205
Pavillard V, Kherfellah D, Richard S, Robert J, Montaudon D . Effects of the combination of camptothecin and doxorubicin or etoposide on rat glioma cells and camptothecin-resistant variants Br J Cancer 2001 85: 1077–1083
de Jong S, Zijlstra JG, De Vries EG, Mulder NH . Reduced DNA topoisomerase II activity and drug-induced DNA cleavage activity in an adriamycin-resistant human small cell lung carcinoma cell line Cancer Res 1990 50: 304–309
Zijlstra JG, De Vries EG, Mulder NH . Multifactorial drug resistance in an adriamycin-resistant human small cell lung carcinoma cell line Cancer Res 1987 47: 1780–1784
Liliemark EK, Liliemark J, Pettersson B, Gruber A, Bjorkholm M, Peterson C . In vivo accumulation of etoposide in peripheral leukaemic cells in patients treated for acute myeloblastic leukaemia; relation to plasma concentration and protein binding Leuk Lymphoma 1993 10: 323–328
Zhou R, Frostvik-Stolt M, Liliemark E . Determination of etoposide in human plasma and leukemic cells by high-performance liquid chromatography with electrochemical detection J Chromatogr B Biomed Sci Appl 2001 757: 135–141
Dubrez L, Goldwasser F, Genne P, Pommier Y, Solary E . The role of cell cycle regulation and apoptosis triggering in determining the sensitivity of leukaemic cells to topoisomerase I and II inhibitors Leukemia 1995 9: 1013–1024
Jia L, Patwari Y, Srinivasula SM, Newland AC, Fernandes-Alnemri T, Alnemri ES, Kelsey SM . Bax translocation is crucial for the sensitivity of leukaemic cells to etoposide-induced apoptosis Oncogene 2001 20: 4817–4826
Oltvai ZN, Milliman CL, Korsmeyer SJ . Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death Cell 1993 74: 609–619
Reed JC . Double identity for proteins of the bcl-2 family Nature 1997 387: 773–776
Kondo S, Barna BP, Kondo Y, Tanaka Y, Casey G, Liu J, Morimura T, Kaakaji R, Peterson JW, Werbel B, Barnett GH . WAF1/CIP1 increases the susceptibility of p53 non-functional malignant glioma cells to cisplatin-induced apoptosis Oncogene 1996 13: 1279–1285
Burger H, Nooter K, Boersma AW, Kortland CJ, Stoter G . Lack of correlation between cisplatin-induced apoptosis, p53 status and expression of bcl-2 family proteins in testicular germ cell tumour cell lines Int J Cancer 1997 73: 592–599
Wang GQ, Gastman BR, Wieckowski E, Goldstein LA, Gambotto A, Kim TH, Fang B, Rabinovitz A, Yin XM, Rabinowich H . A role for mitochondrial Bak in apoptotic response to anticancer drugs J Biol Chem 2001 276: 34307–34317
Pinto AL, Lippard SJ . Binding of the antitumor drug cisdiamminedichloro-platinum (cisplatin) to DNA Biochim Biophys Acta 1985 780: 167–180
Sumner AT, de la Torre J, Stuppia L . The distribution of genes on chromosomes: a cytological approach J Mol Evol 1993 37: 117–122
Felix CA . Secondary leukaemias induced by topoisomerase-targeted drugs Biochim Biophys Acta 1998 1400: 233–255
Felix CA . Leukemias related to treatment with DNA topoisomerase II inhibitors Med Pediatr Oncol 2001 36: 525–535
Melixetian MB, Beryozkina EV, Pavlenko MA, Grinchuk TM . Altered expression of DNA-topoisomerase IIa is associated with increased rate of spontaneous polyplodisation in etoposide resistant K562 cells Leuk Res 2000 24: 831–837
Acknowledgements
We thank Margaret Burridge and Deborah Lillington for assistance with analysing and interpreting the cytogenetic results.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, W., Oakley, P. & Joel, S. Exposure to low concentrations of etoposide reduces the apoptotic capability of leukaemic cell lines. Leukemia 16, 1705–1712 (2002). https://doi.org/10.1038/sj.leu.2402621
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402621
Keywords
This article is cited by
-
Simple and cost-effective liquid chromatography-mass spectrometry method to measure dabrafenib quantitatively and six metabolites semi-quantitatively in human plasma
Analytical and Bioanalytical Chemistry (2017)
-
DNA-AP sites generation by Etoposide in whole blood cells
BMC Cancer (2009)
-
The relative activity of cisplatin, oxaliplatin and satraplatin in testicular germ cell tumour sensitive and resistant cell lines
Cancer Chemotherapy and Pharmacology (2009)
-
The schedule-dependent effects of etoposide in leukaemic cell lines: a function of concentration and duration
Cancer Chemotherapy and Pharmacology (2003)