Abstract
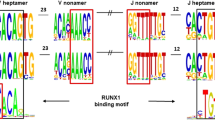
T(14;18) chromosomal translocation is assumed to result from illegitimate rearrangement between the BCL2 proto-oncogene and the IGH locus during the DH to JH joining phase of V(D)J recombination in early B cells. Analysis of the breakpoint junctions suggests that translocation derives from the fusion between normal V(D)J recombination intermediates at the IGH locus and non-V(D)J-mediated broken-ends at the BCL2 locus. So far, BCL2 broken-ends have only been observed fused to coding-ends, raising questions concerning the molecular constraints of the illegitimate joining process. Using a combination of genome walking and long-range PCR assays, we describe in this report that in 4.5% (2/44) of the t(14;18), one of the BCL2 broken-ends is fused to a signal-end. The formation of these JHRSS/BCL2 junctions provides direct evidence that BCL2 broken-ends are capable of joining to both products of V(D)J recombination, suggesting their presence in the RAG-mediated post-cleavage complex. In addition, junctions generated by this alternative end-joining do not involve deletion of the chromosome 14 intervening sequences generally lost in the standard translocation, providing a unique opportunity to investigate the rearrangement status of this region in the translocated IGH allele. In both cases, a DJH rearrangement could be detected 5′ of the JH-RSS/BCL2 junction. These findings, together with the previously reported bias towards the most external DH and JH segments in standard breakpoints, strongly suggest that t(14;18) preferentially occurs during an attempted secondary DH to JH rearrangement. This unusual and restricted window of differentiation opens intriguing questions concerning the etiology of the translocation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Cleary ML, Sklar J . Nucleotide sequence of a t(14;18) chromosomal breakpoint in follicular lymphoma and demonstration of a breakpoint-cluster region near a transcriptionally active locus on chromosome 18 Proc Natl Acad Sci USA 1985 82: 7439–7443
Bakhshi A, Wright JJ, Graninger W, Seto M, Owens J, Cossman J, Jensen JP, Goldman P, Korsmeyer SJ . Mechanism of the t(14;18) chromosomal translocation: structural analysis of both derivative 14 and 18 reciprocal partners Proc Natl Acad Sci USA 1987 84: 2396–2400
Wyatt RT, Rudders RA, Zelenetz A, Delellis RA, Krontiris TG . BCL2 oncogene translocations is mediated by a chi-like consensus J Exp Med 1992 175: 1575–1588
Jaeger U, Boecskoer S, Le T, Mitterbauer G, Bolz I, Chott A, Kneba M, Mannhalter C, Nadel B . Follicular lymphomas'BCL2/IGH junctions contain templated nucleotide insertions: novel insights into the mechanism of t(14;18) translocation Blood 2000 95: 3520–3529
Tsujimoto Y, Louie E, Bashir MM, Croce CM . The reciprocal partners of both the t(14;18) and the t(11;14) translocations involved in B-cell neoplasms are rearranged by the same mechanism Oncogene 1988 2: 347–351
Cotter F, Price C, Zucca E, Young BD . Direct sequence analysis of the 14q+ and 18q− chromosome junctions in follicular lymphoma Blood 1990 76: 131–135
Yamada M, Wasserman R, Reichard BA, Shane S, Caton AJ, Rovera G . Preferential utilization of specific immunoglobulin heavy chain diversity and joining segments in adult human peripheral blood B lymphocytes J Exp Med 1991 173: 395–407
Sanz I . Multiple mechanisms participate in the generation of diversity of human H chain CDR3 regions J Immunol 1991 147: 1720–1729
Milili M, Schiff C, Fougereau M, Tonnelle C . The VDJ repertoire expressed in human preB cells reflects the selection of bona fide heavy chains Eur J Immunol 1996 26: 63–69
Corbett SJ, Tomlinson IM, Sonnhammer ELL, Buck D, Winter G . Sequence of the human immunoglobulin diversity (D) segment locus: a systematic analysis provides no evidence for the use of DIR segments, inverted D segments, ‘minor’ D segments or D–D recombination J Mol Biol 1997 270: 587–597
Buchonnet G, Lenain P, Ruminy P, Lepretre S, Stamatoullas A, Parmentier F, Jardin F, Duval C, Tilly H, Bastard C . Characterisation of BCL2-JH rearrangements in follicular lymphoma: PCR detection of 3′ BCL2 breakpoints and evidence of a new cluster Leukemia 2000 14: 1563–1569
Lewis S, Gifford A, Baltimore D . DNA elements are asymmetrically joined during the site-specific recombination of kappa immunoglobulin genes Science 1985 228: 677–685
Zhu C, Bogue MA, Lim DS, Hasty P, Roth DB . Ku86-deficient mice exhibit severe combined immunodeficiency and defective processing of V(D)J recombination intermediates Cell 1996 86: 379–389
Lieber MR, Hesse JE, Mizuuchi K, Gellert M . Lymphoid V(D)J recombination: nucleotide insertion at signal joints as well as coding joints Proc Natl Acad Sci USA 1988 85: 8588–8592
Tycko B, Coyle H, Sklar J . Chimeric gamma-delta signal joints. Implications for the mechanism and regulation of T cell receptor gene rearrangement J Immunol 1991 147: 705–713
Garcia IS, Kaneko Y, Gonzalez-Sarmiento R, Campbell K, White L, Boehm T, Rabbitts TH . A study of chromosome 11p13 translocations involving TCR beta and TCR delta in human T cell leukaemia Oncogene 1991 6: 577–582
Tycko B, Reynolds TC, Smith SD, Sklar J . Consistent breakage between consensus recombinase heptamers of chromosome 9 DNA in a recurrent chromosomal translocation of human T cell leukemia J Exp Med 1989 169: 369–377
Fugmann SD, Lee AI, Shockett PE, Villey IJ, Schatz DG . The RAG proteins and V(D)J recombination: complexes, ends, and transposition Annu Rev Immunol 2000 18: 495–527
Lewis SM, Hesse JE, Mizuuchi K, Gellert M . Novel strand exchanges in V(D)J recombination Cell 1988 55: 1099–1107
Lewis SM, Hesse JE . Cutting and closing without recombination in V(D)J joining EMBO J 1991 10: 3631–3639
Schatz DG . Developing B-cell theories Nature 1999 400: 614–615, 617
Nemazee D . Receptor editing in B cells Adv Immunol 2000 74: 89–126
Nagaoka H, Yu W, Nussenzweig MC . Regulation of RAG expression in developing lymphocytes Curr Opin Immunol 2000 12: 187–190
Taki S, Schwenk F, Rajewsky K . Rearrangement of upstream DH and VH genes to a rearranged immunoglobulin variable region gene inserted into the DQ52-JH region of the immunoglobulin heavy chain locus Eur J Immunol 1995 25: 1888–1896
Papavasiliou F, Casellas R, Suh H, Qin XF, Besmer E, Pelanda R, Nemazee D, Rajewsky K, Nussenzweig MC . V(D)J recombination in mature B cells: a mechanism for altering antibody responses Science 1997 278: 298–301
Sleckman BP, Gorman JR, Alt FW . Accessibility control of antigen-receptor variable-region gene assembly: role of cis-acting elements Annu Rev Immunol 1996 14: 459–481
Acknowledgements
We are grateful to Ann J Feeney and Susanna M Lewis for comments on the manuscript. This work was supported by a grant for the Interdisciplinary Cooperation Project (ICP) from the University of Vienna, and a grant from the Fonds zur Foerderung der Wissenschaftlichen Forschung (FWF P-13984GEN).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Marculescu, R., Le, T., Böcskör, S. et al. Alternative end-joining in follicular lymphomas’ t(14;18) translocation. Leukemia 16, 120–126 (2002). https://doi.org/10.1038/sj.leu.2402324
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402324