Abstract
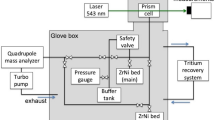
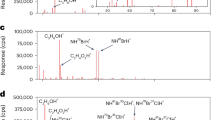
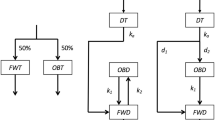
IN the past few years deuterated compounds have become readily available and are in wide use, especially as solvents for NMR spectroscopy. D. Arigoni has informed us (personal communication) that heavy water (D2O) can contain appreciable quantities of tritium. It has been known for some time that tritium is present in D2O obtained as a residue after the prolonged electrolysis of natural water1. But most users of deuterium compounds seem to be unaware of the presence of tritium, often in amounts which require a licence for possession. Those using tritium as a tracer should be cautioned regarding the potential contamination resulting from the use of D2O and other deuterated compounds. We have measured the tritium content of several commercial deuterated compounds which were in routine use in our laboratory, and the results are summarized in Table 1. All the compounds had a nominal deuterium content >99%.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Grosse, A. V., Johnston, W. H., Wolfgang, R. L., and Libby, W. F., Science, 113, 1 (1951).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
LEETE, E., CHEDEKEL, M. Tritium in Deuterated Compounds. Nature 236, 166–167 (1972). https://doi.org/10.1038/236166c0
Received:
Issue Date:
DOI: https://doi.org/10.1038/236166c0
This article is cited by
-
Tritium Contamination of Deuterium Compounds
Nature (1973)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.