Abstract
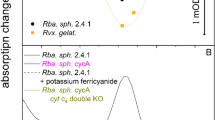
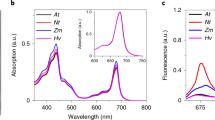
ISOLATED chloroplasts retain most of their structural and physico-chemical properties. Unpublished experiments carried out in this laboratory have shown that the pigments chlorophyll a and β-carotene are held very close together; both are partly removed from the chloroplast by mixtures of ethyl alcohol and water. They are removed in the same proportions and the intensity of photosynthesis is determined from the quantities remaining. Chlorophyll b is more soluble than chlorophyll a and the quantity left after the treatment is not directly related to the intensity of photosynthesis. Reducing system I (β-carotene–ferredoxin–TPN–TPNH)1–3 is also affected by the surrounding solution. Specific quantities of ferredoxin and TPNH are removed by water. After careful preparation in small volumes of solution, however, so much ferredoxin and TPNH is usually left that optimal photosynthesis may occur in red light for 5–10 min. An oxidizing system1 consisting of the cytochromes f, b3 and b6 is also well constructed, but its connexion with chlorophyll (the boundary between cytochrome f and chlorophyll) is more sensitive, and portions of the cytochrome system are frequently removed from the rest of the chloroplast during grinding or sonication. The intensity of photosynthesis then decreases by a proportional extent. Careful isolation in small quantities of liquid leaves most of the chloroplasts untouched and they can then be used for the experiments. The most obvious difference between chloroplasts in whole cells and isolated chloroplasts is that the isolated chloroplasts produced strongly reduced oxygen. Isolated chloroplasts with no additions only occasionally produce limited amounts of oxygen. In most cases, however, some oxygen is absorbed. It is known that isolated chloroplasts are cut off from the metabolic system of the cytoplasm and that they do not respire in the dark. A fundamental difference between chloroplasts in whole cells and isolated chloroplasts is therefore the inability or very small capacity of the latter to produce oxygen. According to the Hill reaction for intense oxygen production an oxidant has to be added; for example, quinone or potassium ferricyanide, both substances with a positive oxidation-reduction potential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lundegårdh, H., Physiol. Plant., 21, 148 (1968).
Lundegårdh, H., Proc. US Nat. Acad. Sci., 59, 293 (1968).
Lundegårdh, H., Nature, 216, 981 (1967).
Lundegårdh, H., Plant Physiology (trans. by Irvine, F. M.; edit. by James, W. O.) (Oliver and Boyd, Edinburgh and London, 1966).
La Photosynthèse, Gif-sur-Yvette, et Saclay, July 1962 (editions Du Centre National de la Recherche Scientifique, Paris, 1963).
Ruhland, W., Encyclopaedia of Plant Physiology, 5, Parts 1 and 2. (The Assimilation of Carbon Dioxide) (Springer-Verlag, Berlin-Göttingen-Heidelberg, 1960).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
LUNDEGÅRDH, H. Relative Quantum Efficiency of Photosynthetic Oxygen Production in Chloroplasts of Spinach. Nature 221, 280–281 (1969). https://doi.org/10.1038/221280a0
Received:
Issue Date:
DOI: https://doi.org/10.1038/221280a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.