Abstract
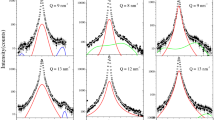
IT is well known1 that for a salt solution the relationship between adiabatic compressibility, β, and molarity, c, is given by the expression:  Recently2, the variation of compressibility with concentration, for aqueous solutions of various inorganic sulphates (lithium sulphate, magnesium sulphate, sodium sulphate, potassium sulphate, etc.), was reported; differences in the behaviour of each solution were related to differences in the cations—as the sulphate ion was common to all, the differences were considered to be due to differences in the ionic radius of the cations.
Recently2, the variation of compressibility with concentration, for aqueous solutions of various inorganic sulphates (lithium sulphate, magnesium sulphate, sodium sulphate, potassium sulphate, etc.), was reported; differences in the behaviour of each solution were related to differences in the cations—as the sulphate ion was common to all, the differences were considered to be due to differences in the ionic radius of the cations.
Similar content being viewed by others
Article PDF
References
Vigoreux, A., Ultrasonics, (John Wiley and Sons, 1960).
Marks, G. W., J. Acoust. Soc. Amer., 31, 7, 936 (1959).
Pauling, L., The Chemical Bond, (1960).
Sette, D., La Ricerca Scientifica, 25, 576 (1955).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
FITTIPALDI, F. Ultrasonic Absorption in Aqueous Solutions of Certain Inorganic Sulphates. Nature 210, 835 (1966). https://doi.org/10.1038/210835a0
Issue Date:
DOI: https://doi.org/10.1038/210835a0
This article is cited by
-
X-ray study of water and aqueous solutions of manganese, zinc, and copper sulfates
Journal of Structural Chemistry (1972)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.