Abstract
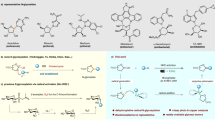
THE teratogenic activity of thalidomide is now well known, but the mechanism by which the drug produces foetal abnormalities is obscure. Recent investigations in this laboratory1 on the relationship between embryotoxic activity and chemical structure in a series of compounds related to thalidomide (α-phthalimidoglutarimide) suggested that the phthalimide portion of the molecule is important in determining teratogenic activity. Replacement of the phthalimide ring of thalidomide by other ring systems such as those of succinimide and hexahydrophthalimide was found to lead to the loss of embryotoxic activity in tho rabbit. However, replacement of the glutarimide ring of thalidomide by other groups did not in every case lead to complete loss of embryopathic activity, for 4-phthalimidobutyramide, phthalimidobenzene and 2-phthalimidoglutaric acid anhydride were still embryotoxic in rabbits but much less so than thalidomide. These observations suggested that an N-substituted phthalimide structure was essential for embryotoxic activity. However, the exact structural requirements in the N-substituent for embryotoxicity are not yet clear, but, so far as is known at present, optimum embryotoxic activity is found when the N-substituent is α-glutarimide as in thalidomide itself.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fabro, S., Schumacher, H., Smith, R. L., and Williams, R. T., Life Sci., 3, 987 (1964).
Fabro, S., Schumacher, H., Smith, R. L., Stagg, R. B. L., and Williams, R. T., Biochem. J., 90, 5–6P (1964).
Drew, H. D. K., and Hatt, H. H., J. Chem. Soc., 16 (1937).
Green, J. N., and Benson, B. C., J. Pharm. Pharmacol., 13, 117T (1961).
Schumacher, H., Smith, R. L., and Williams, R. T., Brit. J. Pharmacol., 25, 324 (1965).
Schumacher, H., Smith, R. L., Stagg, R. B. L., and Williams, R. T., Pharm. Act. Helv., 39, 394 (1964).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
FABRO, S., SMITH, R. & WILLIAMS, R. Thalidomide as a Possible Biological Acylating Agent. Nature 208, 1208–1209 (1965). https://doi.org/10.1038/2081208a0
Issue Date:
DOI: https://doi.org/10.1038/2081208a0
This article is cited by
-
Thalidomide-induced limb abnormalities in a humanized CYP3A mouse model
Scientific Reports (2016)
-
Embryotoxicit�t und Teratogenit�t von Derivaten des 1, 3-Indandion
Archiv f�r Toxikologie (1975)
-
The hydrolysis of polyimides
Experientia (1973)
-
Accumulation of calcium and phosphate stimulated by carboxylic antibiotics into mitochondria
Journal of Bioenergetics (1972)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.