Abstract
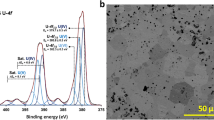
IN a recent communication by Sasaki et al.1 attention is directed to the use of the carbides of uranium, plutonium and thorium as nuclear fuels and to the problems associated with processing and recovery of the irradiated fuel. It is revealed that in the acid leaching of thorium carbide (ThC2) with nitric acid of concentrations below 4 N gaseous hydrocarbons are evolved, whereas at concentrations above 4 N organic material is produced, and evidence is presented that this organic matter does not have a C—H bond as indicated by the results of infra-red analysis. The formation of organic material during the acid leaching of uranium monocarbide has also been observed by Simpson et al.2 and the presence of this could constitute a hazard in the uranium recovery process, although its formation may be avoided by prior hydrolysis of the carbide in water and subsequent dissolution of the resulting oxides in nitric acid3.
Similar content being viewed by others
Article PDF
References
Sasaki, Y., Ichikawa, F., Imai, H., and Uruno, S., Nature, 195, 267 (1962).
Simpson, A. M., and Heath, B. A., U.K. Atomic Energy Auth. (private communication).
Hartley, K., and Maclennan, G., British Pat. 5682/58 (April 1958).
Healy, T. V., and McKay, H. A. C., Rec. Trav. Chim., 75, 730 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PAUSON, P., McLEAN, J. & CLELLAND, W. Acid Leaching of Uranium and Thorium Carbides. Nature 197, 1200 (1963). https://doi.org/10.1038/1971200a0
Issue Date:
DOI: https://doi.org/10.1038/1971200a0
This article is cited by
-
Uranium carbide dissolution in nitric acid: speciation of organic compounds
Journal of Radioanalytical and Nuclear Chemistry (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.