Abstract
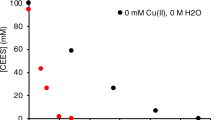
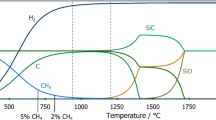
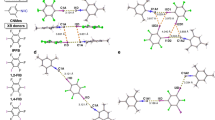
A NUMBER of methods are available for the preparation of trichlorosilane (SiHCl3), in the laboratory, for example, by the action of dry hydrochloric acid gas on silicon1,2 and by the reaction of hydrochloric acid gas and metallic silicides3–7, such as magnesium silicide, iron silicide, or 25 per cent copper silicide. Trichlorosilane has also been prepared by passing a mixture of halosilanes and excess of hydrogen or hydrogen-halide over aluminium, zinc, or magnesium8, or by halogenation of monosilane (SiH4), with hydrochloric acid gas in presence of aluminium chloride as a catalyst9.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Buff, H., and Wöhler, F., Ann. Chem., 104, 94 (1857).
Friedel, C., and Ladenburg, A., Ann. Chem., 143, 118 (1867).
Gattermann, L., Ber., 22, 190 (1889).
Brauer, G., Handbuch der präparativen anorg. Chemie (F. Enke, Verlag, Stuttgart, 1954).
Warren, F., Ber., 22, 657 (1889).
Taylor, A. G., and Walden, G., J. Amer. Chem. Soc., 66, 842 (1944).
Ruff, O., and Albert, K., Ber., 38, 2222 (1905).
Hurd, R., J. Amer. Chem. Soc., 67, 1545 (1954). U.S. Patent 2.406.605.
Stock, D., Hydrides of Boron and Silicon (Cornell Univ. Press, Ithaca, New York).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DUDANI, P., PLUST, H. A Simplified Method for Preparation of Very Pure Silicochloroform. Nature 194, 85–86 (1962). https://doi.org/10.1038/194085a0
Issue Date:
DOI: https://doi.org/10.1038/194085a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.