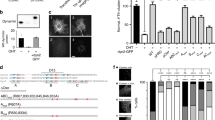
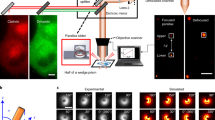
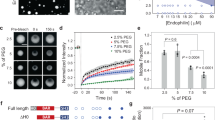
Abstract
Dynamin is a GTP-hydrolysing protein that is an essential participant in clathrin-mediated endocytosis by cells. It self-assembles into ‘collars’ in vitro which also form in vivo at the necks of invaginated coated pits. This self-assembly stimulates dynamin's GTPase activity and it has been proposed that dynamin hydrolyses GTP in order to generate the force needed to sever vesicles from the plasma membrane. A mechanism is now described in which self-assembly of dynamin is coordinated by a domain of dynamin with a GTPase-activating function. Unexpectedly, when dynamin mutants defective in self-assembly-stimulated GTPase activity are overexpressed, receptor-mediated endocytosis is accelerated. The results indicate that dynamin, like other members of the GTPase superfamily, functions as a molecular regulator in receptor-mediated endocytosis, rather than as a force-generating GTPase.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Schmid, S. L. Clathrin-coated vesicle formation and protein sorting: An integrated process. Annu. Rev. Biochem. 66, 511–548 (1997).
Schmid, S. L., McNiven, M. A. & De Camilli, P. Dynamin and its partners: A progress report. Curr. Opin. Cell Biol. 10, 504–512 (1998).
van der Bliek, A. M. Functional diversity in the dynamin family. Trends Cell Biol. 9, 96–102 (1999).
Muhlberg, A. B., Warnock, D. E. & Schmid, S. L. Domain structure and intramolecular regulation of dynamin GTPase. EMBO J. 16, 6676–6683 (1997).
Hinshaw, J. E. & Schmid, S. L. Dynamin self assembles into rings suggesting a mechanism for coated vesicle budding. Nature 374, 190–192 (1995).
Warnock, D. E., Hinshaw, J. E. & Schmid, S. L. Dynamin self assembly stimulates its GTPase activity. J.Biol. Chem. 271, 22310–22314 (1996).
Lin, H. C. & Gilman, A. G. Regulation of dynamin I GTPase activity by G protein betagamma subunits and phosphatidylinositol 4,5-bisphosphate. J. Biol. Chem. 271, 27979–27982 (1996).
Tuma, P. L., Stachniak, M. C. & Collins, C. A. Activation of dynamin GTPase by acidic phospholipids and endogenous rat brain vesicles. J. Biol. Chem. 268, 17240–17246 (1993).
Barylko, B. et al. Synergistic activation of dynamin GTPase by Grb2 and phosphoinositides. J. Biol. Chem. 273, 3791–3797 (1998).
Kosaka, T. & Ikeda, K. Possible temperature-dependent blockage of synaptic vesicle recycling induced by a single gene mutation in Drosophila. J. Neurobiol. 14, 207–225 (1983).
Takei, K., McPherson, P. S., Schmid, S. L. & De Camilli, P. Tubular membrane invaginations coated by dynamin rings are induced by GTP-γS in nerve terminals. Nature 374, 186–190 (1995).
Sweitzer, S. & Hinshaw, J. Dynamin undergoes a GTP-dependent conformational change causing vesiculation. Cell 93, 1021–1029 (1998).
Takei, K. et al. Generation of coated intermediates of clathrin-mediated endocytosis on protein-free liposomes. Cell 94, 131–141 (1998).
Warnock, D. E. & Schmid, S. L. Dynamin GTPase, a force-generating molecular switch. Bioessays 18, 885–893 (1996).
Liu, J. P. & Robinson, P. J. Dynamin and endocytosis. Endocrine Rev. 16, 590–607 (1995).
McNiven, M. A. Dynamin: a molecular motor with pinchase action. Cell 94, 151–607 (1998).
Shpetner, H. S. & Vallee, R. B. Dynamin is a GTPase stimulated to high levels of activity by microtubules. Nature 355, 733–735 (1992).
Tuma, P. L. & Collins, C. A. Dynamin forms polymeric complexes in the presence of lipid vesicles. Characterization of chemically crosslinked dynamin molecules. J. Biol. Chem. 270, 26707–26714 (1995).
Schmid, S. L. & Smythe, E. Stage-specific assays for coated pit formation and coated vesicle budding in vitro. J. Cell Biol. 114, 869–880 (1991).
Carter, L. L., Redelmeier, T. E., Woollenweber, L. A. & Schmid, S. L. Multiple GTP-binding proteins participate in clathrin-coated vesicle-mediate endocytosis. J. Cell Biol. 120, 37–45 (1993).
Maeda, K., Nakata, T., Noda, Y., Sato-Yoshitake, R. & Hirokawa, N. Interaction of dynamin with microtubules: its structure and GTPase activity investigated by using highly purified dynamin. Mol. Biol. Cell 3, 1181–1194 (1992).
Mittal, R., Ahmadian, M. R., Goody, R. S. & Wittinghofer, A. Formation of a transition-state analog of the Ras GTPase reaction by Ras-GDP, tetrafluoraluminate and GTPase activation proteins. Science 273, 115–117 (1996).
Carr, J. F. & Hinshaw, J. E. Dynamin assembles into spirals under physiological salt conditions upon the addition of GDP and gamma-phosphate analogues. J. Biol. Chem. 272, 28030–28035 (1997).
Scheffzek, K., Ahmadian, R. & Wittinghofer, A. GTPase-activating proteins: helping hands to complement active site. Trends Biochem. Sci. 23, 257–262 (1998).
Herskovits, J. S., Burgess, C. C., Obar, R. A. & Vallee, R. B. Effects of mutant rat dynamin on endocytosis. J. Cell Biol. 122, 565–578 (1993).
Melen, K. et al. Interferon-induced Mx proteins form oligomers and contain a putative leucine zipper. J. Biol. Chem. 267, 25898–25907 (1992).
Schwemmle, M., Richter, M. F., Hermann, C., Nassar, N. & Staeheli, P. Unexpected structural requirements for GTPase activity of the interferon-induced MxA protein. J. Biol. Chem. 270, 13518–13523 (1995).
Warnock, D. E., Terlecky, L. J. & Schmid, S. L. Dynamin GTPase is stimulated by crosslinking through the C terminal proline rich domain. EMBO J. 14, 1322–1328 (1995).
Gilbert, A., Paccaud, J. P. & Carpentier, J. L. Direct measurement of clathrin-coated vesicle formation using a cell-free assay. J. Cell Sci. 110, 3105–3115 (1997).
Gout, I. et al. The GTPase dynamin binds to and is activated by a subset of SH3 domains. Cell 75, 25–36 (1993).
Foster-Barber, A. & Bishop, J. M. Src interacts with dynamin and synapsin in neuronal cells. Proc. Natl Acad. Sci. USA 95, 4673–4677 (1998).
Earnest, S., Khokhlatchev, A., Albanesi, J. P. & Barylko, B. Phosphorylation of dynamin by ERK2 inhibits the dynamin-microtubule interaction. FEBS Lett. 396, 62–66 (1996).
Fersht, A. Enzyme Structure and Mechanism(Freeman, New York, (1995).
van der Bliek, A. M. et al. Mutations in human dynamin block an intermediate stage in coated vesicle formation. J. Cell Biol. 122, 553–563 (1993).
van der Bliek, A. M. & Meyerowitz, E. M. Dynamin-like protein encoded by the Drosophila shibire gene associated with vesicular traffic. Nature 351, 411–414 (1991).
Chen, M. S. et al. Multiple forms of dynamin are encoded by shibire, a Drosophila gene involved in endocytosis. Nature 351, 583–586 (1991).
Rothman, J. H., Raymond, C. K., Gilbert, T., O'Hara, P. J. & Stevens, T. H. Aputative GTP binding protein homologous to interferon-inducible Mx proteins performs an essential function in yeast protein sorting. Cell 61, 1063–1074 (1990).
Gammie, A. E., Kurihar, K. L., Vallee, R. B. & Rose, M. D. DNM1, a dynamin-related gene, participates in endosomal trafficking in yeast. J. Cell Biol. 130, 553–566 (1995).
Otsuga, D. et al. The dynamin-like GTPase, Dnm1p, controls mitochondrial morphology in yeast. J. Cell Biol. 143, 333–349 (1998).
Aebi, M. et al. cDNA structure and regulation of two interferon-induced human Mx proteins. Mol. Cell. Biol. 9, 5062–5072 (1989).
Gu, X. & Verma, D. P. Phragmoplastin, a dynamin-like protein associated with cell plate formation in plants. EMBO J. 15, 695–704 (1996).
Acknowledgements
We thank H. Damke for her contribution to analysis of dynamin function in vivo, L.M. Fujimoto and F. Simpson for advice and assistance, and J. Walter, W. E. Balch, L. Gerace, H. Damke and S. Newmyer for critically reading the manuscript. S.L.S. was supported by grants from the NIH and is an established investigator of the American Heart Association. S.S. was supported by a postdoctoral fellowship from the American Cancer Society and A.B.M. was supported by a predoctoral fellowship from the American Heart Association.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sever, S., Muhlberg, A. & Schmid, S. Impairment of dynamin's GAP domain stimulates receptor-mediated endocytosis. Nature 398, 481–486 (1999). https://doi.org/10.1038/19024
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/19024
This article is cited by
-
Reversal of cell, circuit and seizure phenotypes in a mouse model of DNM1 epileptic encephalopathy
Nature Communications (2023)
-
Role of actin cytoskeleton in podocytes
Pediatric Nephrology (2021)
-
Reduction in MLKL-mediated endosomal trafficking enhances the TRAIL-DR4/5 signal to increase cancer cell death
Cell Death & Disease (2020)
-
Molecular cloning, GTP recognition mechanism and tissue-specific expression profiling of myxovirus resistance (Mx) protein in Labeo rohita (Hamilton) after Poly I:C induction
Scientific Reports (2019)
-
Conformational dynamics of dynamin-like MxA revealed by single-molecule FRET
Nature Communications (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.