Abstract
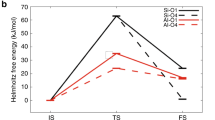
THE chemistry of silicic acid in solution is rather ill-defined1 owing to the extremely weak nature of this acid2 and its tendency to polymerize3,4. As there are many instances where hydrogen peroxide effectively prevents anionic polymerization5,6, an attempt was made to investigate the hydrolysis of silicate ions in the presence of hydrogen peroxide by means of accurate conductivity7, and pH measurements. For comparison, the same technique was applied to an identical series of solutions in the absence of hydrogen peroxide. Fig. 1 shows the results of the investigation on solutions which were 0.01 M in sodium silicate and, in the case of the ‘peroxidized’ system, approximately 0.1 M in hydrogen peroxide; sulphuric acid was 0.498 M.
Similar content being viewed by others
Article PDF
References
Greenberg, S. A., J. Chem. Educ., 36, 218 (1959).
Remy, H., “Treatise on Inorganic Chemistry”, 1, 493 (Elsevier, Amsterdam, 1956).
Thilo, E., Angew. Chem., 63, 201 (1951).
Alexander, G. B., J. Amer. Chem. Soc., 76, 2094 (1954).
Kakabadse, G., and Wilson, H. J., Nature, 180, 861 (1957).
Jahr, K. F., “Fiat Review of German Science 1936–1946”, Pt. 3, 172.
Constable, F. H., and Tegul, S., Nature, 160, 55 (1947).
Kortüm, G., and Bockris, J. O'M., “Textbook of Electrochemistry”, 1, 216 (Elsevier, London, 1951).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KAKABADSE, G., DEWSNAP, J. The Sodium Silicate – Hydrogen Peroxide System. Nature 185, 761 (1960). https://doi.org/10.1038/185761a0
Issue Date:
DOI: https://doi.org/10.1038/185761a0
This article is cited by
-
Siliciumstoffwechsel bei Mikroorganismen
Archiv f�r Mikrobiologie (1965)
-
The Sodium Silicate – Ferrous Hydroxide System
Nature (1961)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.