Summary:
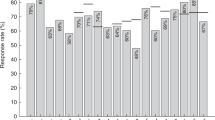
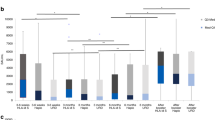
During follow-up after allogeneic stem cell transplantation (SCT), patients frequently lose their immunity to infectious agents such as measles. The aim of this study was to analyze the influence of different factors on measles immunity. In total, 395 patients with a disease-free survival of at least 1 year were included. Measles vaccination was given at 2 years after SCT to children and young adults without chronic GVHD or ongoing immunosuppression. In all, 264 patients had matched sibling donors and 131 either mismatched family or unrelated donors. Totally, 318 patients received bone marrow and 77 peripheral blood stem cells. Overall, 375 patients had undergone myeloablative and 20 nonmyeloablative conditioning. Out of 395 patients, 133 (34%) were seronegative to measles. In multivariate models, younger age or being vaccinated to measles, rather than previous measles disease, before transplantation were risk factors both for becoming seronegative and to have doubtfully protective immunity to measles. Acute GVHD grade II–IV was a risk factor for seronegativity and blood stem cells a risk factor for doubtfully protective immunity. Children and young adults previously immunized to measles have a high risk for becoming vulnerable to a measles infection. Since measles is again circulating in many countries and measles is a serious infection after SCT, vaccination should be considered.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Ljungman P, Lewensohn-Fuchs I, Hammarstrom V et al. Long-term immunity to measles, mumps, and rubella after allogeneic bone marrow transplantation. Blood 1994; 84 (2): 657–663.
Machado CM, Goncalves FB, Pannuti CS et al. Measles in bone marrow transplant recipients during an outbreak in Sao Paulo, Brazil. Blood 2002; 99 (1): 83–87.
Nakano T, Shimono Y, Sugiyama K et al. Clinical features of measles in immunocompromised children. Acta Paediatr Jpn 1996; 38 (3): 212–217.
Kaplan L, Daum R, Smaron M, McCarthy C . Severe measles in immunocompromised patients. JAMA 1992; 267 (9): 1237–1241.
Ringden O, Remberger M, Carlens S et al. Low incidence of acute graft-versus-host disease, using unrelated HLA-A-, HLA-B-, and HLA-DR-compatible donors and conditioning, including anti-T-cell antibodies. Transplantation 1998; 66 (5): 620–625.
Ringdén O, Remberger M, Persson U et al. Similar incidence of graft-versus-host disease using HLA-A, -B and -DR identical unrelated bone marrow donors as with HLA-identical siblings. Bone Marrow Transplant 1995; 15 (4): 619–625.
Ringdén O, Pihlstedt P, Markling L et al. Prevention of graft-versus-host disease with T-cell depletion or cyclosporin and methotrexate. A randomized trial in adult leukemic marrow recipients. Bone Marrow Transplant 1991; 7: 221–226.
Ljungman P, Fridell E, Lönnqvist B et al. Efficacy and safety of vaccination of marrow transplant recipients with a live attenuated measles, mumps, and rubella vaccine. J Infect Dis 1989; 159 (4): 610–615.
Ljungman P . Immunization of transplant recipients. Bone Marrow Transplant 1999; 23 (7): 635–636.
Remberger M, Kumlien G, Aschan J et al. Risk factors for moderate-to-severe chronic graft-versus-host disease after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2002; 8 (12): 674–682.
Ringden O, Labopin M, Bacigalupo A et al. Transplantation of peripheral blood stem cells as compared with bone marrow from HLA-identical siblings in adult patients with acute myeloid leukemia and acute lymphoblastic leukemia. J Clin Oncol 2002; 20 (24): 4655–4664.
Ringden O, Remberger M, Runde V et al. Peripheral blood stem cell transplantation from unrelated donors: a comparison with marrow transplantation. Blood 1999; 94 (2): 455–464.
McSweeney PA, Niederwieser D, Shizuru JA et al. Hematopoietic cell transplantation in older patients with hematologic malignancies: replacing high-dose cytotoxic therapy with graft-versus-tumor effects. Blood 2001; 97 (11): 3390–3400.
Update: global measles control and mortality reduction – worldwide. 1991–2001. MMWR – Morb Mortal Wkly Rep 2003; 52 (20): 471–475.
Maris M, Boeckh M, Storer B et al. Immunologic recovery after hematopoietic cell transplantation with nonmyeloablative conditioning. Exp Hematol 2003; 31 (10): 941–952.
D'Sa S, Peggs K, Pizzey A et al. T- and B-cell immune reconstitution and clinical outcome in patients with multiple myeloma receiving T-cell-depleted, reduced-intensity allogeneic stem cell transplantation with an alemtuzumab-containing conditioning regimen followed by escalated donor lymphocyte infusions. Br J Haematol 2003; 123 (2): 309–322.
Kanra G, Cetin I, Akcoren Z et al. Giant cell pneumonia in a leukemic child in remission: a case report. Turk J Pediatr 2001; 43 (4): 338–341.
Haltia M, Paetau A, Vaheri A et al. Fatal measles encephalopathy with retinopathy during cytotoxic chemotherapy. J Neurol Sci 1977; 32 (3): 323–330.
Breitfeld V, Hashida Y, Sherman F et al. Fatal measles infection in children with leukemia. Lab Invest 1973; 29: 279–281.
Measles epidemic attributed to inadequate vaccination coverage – Campania, Italy. 2002. MMWR – Morb Mortal Wkly Rep 2003; 53 (43): 1044–1047.
Nakajima N, Matsuda T, Ono T et al. Measles outbreak in a suburb of Tokyo, Japan, in 1998–1999. Scand J Infect Dis 2003; 35 (8): 495–497.
McBrien J, Murphy J, Gill D et al. Measles outbreak in Dublin, 2000. Pediatr Infect Dis J 2003; 22 (7): 580–584.
Anonymous. Measles pneumonitis following measles–mumps–rubella vaccination of a patient with HIV infection. MMWR – Morb Mort Weekly Rep 1996; 45 (28): 603–606.
CDC. Guidelines for preventing opportunistic infections among hematopoietic stem cell transplant recipients. Recommendations of CDC, the Infectious Disease Society of America, and the American Society of Blood and Marrow Transplantation. MMWR – Morb Mortal Wkly Rep 2000; 49 (RR-10): 1–125.
Acknowledgements
This study was supported by the Swedish Cancer Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ljungman, P., Aschan, J., Barkholt, L. et al. Measles immunity after allogeneic stem cell transplantation; influence of donor type, graft type, intensity of conditioning, and graft-versus host disease. Bone Marrow Transplant 34, 589–593 (2004). https://doi.org/10.1038/sj.bmt.1704634
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704634
Keywords
This article is cited by
-
Humoral immune response to tick-borne encephalitis vaccination in allogeneic blood and marrow graft recipients
npj Vaccines (2020)
-
Impfen bei Immundefizienz
Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz (2020)
-
Pretransplant vaccinations in allogeneic stem cell transplantation donors and recipients: an often-missed opportunity for immunoprotection?
Bone Marrow Transplantation (2015)
-
Live (vaccines) from New York
Bone Marrow Transplantation (2013)
-
Vaccination of stem cell transplant recipients: recommendations of the Infectious Diseases Working Party of the EBMT
Bone Marrow Transplantation (2005)