Abstract
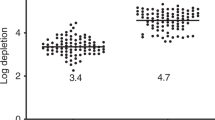
We have investigated the feasibility and efficacy of large-scale T cell depletion from granulocyte colony- stimulating factor (G-CSF) mobilized peripheral blood stem cells (PBSC). The method is based on the use of a CD3 antibody conjugated to magnetic microbeads and magnetic activated cell sorting (Clinimacs). A total of eight large-scale experiments were performed. In four experiments, CD3+ T cells were depleted from PBSC obtained from volunteers mobilized with G-CSF whereas, in four experiments, T cells were depleted from PBSC from stem cell donors, in which the CD34+ stem cells had been removed for allogeneic transplantation by positive selection prior to T cell depletion. The mean number of processed mononuclear cells (MNCs) was 3.3 × 1010 (range 1.5 × 1010–5.1 × 1010) with a mean T cell proportion of 35.8% (range 16.7–64.0%). After T cell depletion, the percentage of contaminating T cells was 0.15% (range 0.01–1.01%) with a mean log10 depletion of 3.4 (range 2.8–4.1). The mean recovery of CD3-negative MNCs after depletion was 76% (range 52–100%). The mean recovery of CD34+ stem cells in the four evaluable experiments was 82% (range 75–92%). In vitro colony assays and in vivo NOD/SCID repopulation assays showed that this large-scale T cell depletion method has no negative impact on the function of the hematopoietic precursor cells. Therefore, we conclude that this T cell depletion method is a valuable tool for further graft engineering strategies involving mobilized PBSCs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Przepiorka D, Ippoliti C, Khouri I et al. Allogeneic transplantation for advanced leukemia: improved short-term outcome with blood stem cell grafts and tacrolimus Transplantation 1996 62: 1806 1810
Russell JA, Brown C, Bowen T et al. Allogeneic blood cell transplants for haematological malignancy: preliminary comparison of outcomes with bone marrow transplantation Bone Marrow Transplant 1996 17: 703 708
Bensinger WI, Clift R, Martin P et al. Allogeneic peripheral blood stem cell transplantation in patients with advancedhematologic malignancies: a retrospective comparison withmarrow transplantation Blood 1996 88: 2794 2800
Schmitz N, Bacigalupo A, Hasenclever D et al. Allogeneic bone marrow transplantation vs filgrastim-mobilised peripheral blood progenitor cell transplantation in patients with early leukaemia: first results of a randomised multicentre trial of the European Group for Blood and Marrow Transplantation Bone Marrow Transplant 1998 21: 995 1003
Zaucha JM, Gooley T, Bensinger WI et al. CD34 cell dose in granulocyte colony–stimulating factor-mobilized peripheral blood mononuclear cell grafts affects engraftment kinetics and development of extensive chronic graft-versus-host disease after human leukocyte antigen-identical sibling transplantation Blood 2001 98: 3221 3227
Handgretinger R, Klingebiel T, Lang P et al. Megadose transplantation of purified peripheral blood CD34(+)progenitor cells from HLA-mismatched parental donors in children Bone Marrow Transplant 2001 27: 777 783
Aversa F, Tabilio A, Velardi A et al. Treatment of high-risk acute leukemia with T cell-depleted stem cells from related donors with one fully mismatched HLA haplotype New Engl J Med 1998 339: 1186 1193
Gilmore MJ, Patterson J, Ivory K et al. Standardization of T-cell depletion in HLA matched bone marrow transplantation Br J Haematol 1986 64: 69 75
Champlin RE, Passweg JR, Zhang MJ et al. T cell depletion of bone marrow transplants for leukemia from donors other than HLA-identical siblings: advantage of T cell antibodies with narrow specificities Blood 2000 95: 3996 4003
Schumm M, Lang P, Taylor G et al. Isolation of highly purified autologous and allogeneic peripheral CD34+ cells using the CliniMACS device J Hematother 1999 8: 209 218
Beelen DW, Peceny R, Elmaagacli A et al. Transplantation of highly purified HLA-identical sibling donor peripheral blood CD34+ cells without prophylactic post-transplant immunosuppression in adult patients with first chronic phase chronic myeloid leukemia: results of a phase II study Bone Marrow Transplant 2000 26: 823 829
Lang P, Handgretinger R, Schumm M et al. Transplantation of purified peripheral CD34+ stem cells from unrelated donors in children: Effective prevention of GVHD Blood 1999 94: (Suppl. 1) 667a
Kato S, Yabe H, Yasui M et al. Allogeneic hematopoietic transplantation of CD34+ selected cells from an HLA haplo-identical related donor. A long-term follow-up of 135 patients and a comparison of stem cell source between the bone marrow and the peripheral blood Bone Marrow Transplant 2000 26: 1281 1290
Eyrich M, Lang P, Lal S et al. A prospective analysis of the pattern of immune reconstitution in a paediatric cohort following transplantation of positively selected human leucocyte antigen-disparate haematopoietic stem cells from parental donors Br J Haematol 2001 114: 422 432
Schreiber KL, Forman J . Effect of graft-versus-host disease on anti-tumor immunity J Immunol 1990 144: 2018 2026
Switzer GE, Goycoolea JM, Dew MA et al. Donating stimulated peripheral blood stem cells vs bone marrow: do donors experience the procedures differently? Bone Marrow Transplant 2001 27: 917 923
Hooks MA, Wade CS, Millikan WJ Jr . Muromonab CD-3: a review of its pharmacology, pharmacokinetics, and clinical use in transplantation Pharmacotherapy 1991 11: 26 37
Keever-Taylor CA, Craig A, Molter M et al. Complement-mediated T-cell depletion of bone marrow: comparison of T10B9.1A-31 and Muronomab-Orthoclone OKT3 Cytotherapy 2001 3: 467 481
Murphy WJ, Koh CY, Raziuddin A et al. Immunobiology of natural killer cells and bone marrow transplantation: merging of basic and preclinical studies Immunol Rev 2001 181: 279 289
Zeis M, Uharek L, Glass B et al. Allogeneic NK cells as potent antileukemic effector cells after allogeneic bone marrow transplantation in mice Transplantation 1995 59: 1734 1736
Bonig H, Korholz D, Lex C et al. Monocyte deactivation and its reversal in a patient with chemotherapy-induced leukopenia and severe systemic infection Med Pediatr Oncol 2000 34: 39 42
Sato K, Hida S, Takayanagi H et al. Antiviral response by natural killer cells through TRAIL gene induction by IFN-alpha/beta Eur J Immunol 2001 31: 3138 3146
Biron CA, Brossay L . NK cells and NKT cells in innate defense against viral infections Curr Opin Immunol 2001 13: 458 464
Dokun AO, Chu DT, Yang L et al. Analysis of in situ NK cell responses during viral infection J Immunol 2001 167: 5286 5293
Osawa M, Hanada K, Hamada H, Nakauchi H . Long-term lymphohematopoietic reconstitution by a single CD34-low/negative hematopoietic stem cell Science 1996 273: 242 245
Zanjani ED, Almeida-Porada G, Livingston AG et al. Human bone marrow CD34- cells engraft in vivo and undergo multilineage expression that includes giving rise to CD34+ cells Exp Hematol 1998 26: 353 360
Bhatia M, Bonnet D, Murdoch B et al. A newly discovered class of human hematopoietic cells with SCID-repopulating activity Nat Med 1998 4: 1038 1045
Bonnet D . Normal and leukemic CD34-negative human hematopoietic stem cells Rev Clin Exp Hematol 2001 5: 42 61
de Wynter EA, Buck D, Hart C et al. CD34+AC133+ cells isolated from cord blood are highly enriched in long-term culture-initiating cells, NOD/SCID-repopulating cells and dendritic cell progenitors Stem Cells 1998 16: 387 396
van Esser JW, van der HB, Meijer E et al. Epstein–Barr virus (EBV) reactivation is a frequent event after allogeneic stem cell transplantation (SCT) and quantitatively predicts EBV-lymphoproliferative disease following T-cell-depleted SCT Blood 2001 98: 972 978
Heslop HE, Rooney CM . Adoptive cellular immunotherapy for EBV lymphoproliferative disease Immunol Rev 1997 157: 217 222
Faye A, Quartier P, Reguerre Y et al. Chimaeric anti-CD20 monoclonal antibody (rituximab) in post-transplant B-lymphoproliferative disorder following stem cell transplantation in children Br J Haematol 2001 115: 112 118
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gordon, P., Leimig, T., Mueller, I. et al. A large-scale method for T cell depletion: towards graft engineering of mobilized peripheral blood stem cells. Bone Marrow Transplant 30, 69–74 (2002). https://doi.org/10.1038/sj.bmt.1703619
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703619
Keywords
This article is cited by
-
Low-dose anti-thymocyte globulin reduce severe acute and chronic graft-versus-host disease after allogeneic stem cell transplantation
Journal of Cancer Research and Clinical Oncology (2017)
-
CD6 as a Therapeutic Target in Autoimmune Diseases: Successes and Challenges
BioDrugs (2013)
-
New strategies for haploidentical transplantation
Pediatric Research (2012)
-
Haploidentical SCT in children: an update and future perspectives
Bone Marrow Transplantation (2008)
-
Mobilization, harvesting and selection of peripheral blood stem cells in patients with autoimmune diseases undergoing autologous hematopoietic stem cell transplantation
Bone Marrow Transplantation (2007)