Abstract
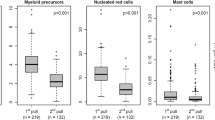
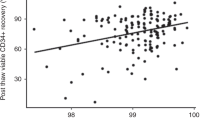
The number of viable precursor cells actually reinfused into patients after high-dose chemotherapy is one of the most clinically important variables determining graft success or failure. A modified, previously described flow cytometric method based on annexin V staining was therefore applied to assess the degree of apoptosis and necrosis in cryopreserved PBPC concentrates from patients with malignant diseases. Twenty-two samples of unmanipulated cryopreserved PBPC concentrates were analyzed by flow cytometry. The samples were triple-stained with anti-CD34 PE, annexin V-FITC and actinomycin D, which enabled the separation of viable, early apoptotic and late apoptotic/necrotic CD34+ precursor cells. Apotosis and necrosis were also measured in the total cell population of the concentrates. Eighty-one percent (range 49–97) of the CD34+ cells were viable, while 7% (range 1–15) were early apoptotic and 12% (range 2–36) were late apoptotic/necrotic after freeze/thaw. There was no difference in apoptosis and necrosis in CD34+ cells harvested from mildly pretreated patients with multiple myeloma and heavily pre-treated patients with non-Hodgkin's lymphoma. Apoptosis and necrosis were higher in the total mature cell population of the concentrates. Thirty-two percent (range 7–69) of the cells were apoptotic and 33% (range 12–60) were necrotic. We conclude that flow cytometric analysis of annexinV/actinomycin D binding in PBPC concentrates is a simple technique that can give additional information of the viability status of the cells post thaw. The present study confirms the relative robustness of human CD34+ precursor cells concerning the freeze/thaw procedure, which are carried out in daily clinical practice.
Bone Marrow Transplantation (2002) 29, 165–171. doi:10.1038/sj.bmt.1703346
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
To LB, Haylock DN, Simmons PJ et al. The biology and clinical uses of blood precursor cells Blood 1997 89: 2233 2258
Haas R, Witt B, Mohle R et al. Sustained long-term hematopoiesis after myeloablative therapy with peripheral blood progenitor cell support Blood 1995 85: 3754 3761
Siena S, Bregni M, Di Nicola M et al. Durability of hematopoiesis following autografting with peripheral blood hematopoietic progenitors Ann Oncol 1994 5: 935 941
Lin CK, Kalunta CI, Chen FS et al. Dimethyl sulfoxide suppresses apoptosis in Burkitt's lymphoma cells Exp Cell Res 1995 216: 403 410
De Boer F, Drager AM, Van der WE et al. Changes in L-selectin expression on CD34-positive cells upon cryopreservation of peripheral blood precursor cell transplants Bone Marrow Transplant 1998 22: 1103 1110
Schmidt-Mende J, Hellstrom-Lindberg E, Joseph B et al. Freezing induces artificial cleavage of apoptosis-related proteins in human bone marrow cells J Immunol Meth 2000 245: 91 94
Schuurhuis GJ, Muijen MM, Oberink JW et al. Large populations of non-clonogenic early apoptotic CD34-positive cells are present in frozen–thawed peripheral blood precursor cell transplants Bone Marrow Transplant 2001 27: 487 498
Keeney M, Chin-Yee I, Weir K et al. Single platform flow cytometric absolute CD34+ cell counts based on the ISHAGE guidelines. International Society of Hematotherapy and Graft Engineering Cytometry 1998 34: 61 70
Fadok VA, Voelker DR, Campbell PA et al. Exposure of phosphatidylserine on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages J Immunol 1992 148: 2207 2216
Martin SJ, Reutlingsberger CP, McGahon AJ et al. Early redistribution of plasma membrane phosphatidylserine is a general feature of apoptosis regardless of the initiating stimulus: inhibition by overexpression of Bcl-2 and Abl J Exp Med 1995 82: 1545 1556
Van Engeland M, Nieland LJW, Ramaekers FCS et al. Annexin-V-affinity assay: a review based on an apoptosis detection system based on phosphatidylserine exposure Cytometry 1998 31: 1 9
Vermes I, Haanen C, Reutlingsperger C . Flow cytometry of apoptotic cell death J Immunol Meth 2000 243: 167 190
Andree HA, Reutelingsperger CP, Hauptmann R et al. Binding of vascular anticoagulant alpha (VAC alpha) to planar phospholipid bilayers J Biol Chem 1990 265: 4923 4928
Koopman G, Reutelingsperger CP, Kuijten GA et al. Annexin V for flow cytometric detection of phosphatidylserine expression on B cells undergoing apoptosis Blood 1994 84: 1415 1420
Vermes I, Haanen C, Steffens-Nakken H, Reutelingsperger C . A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled annexin V J Immunol Meth 1995 184: 39 51
Schmidt-Mende J, Hellstrom-Lindberg E, Joseph B et al. Freezing induces artificial cleavage of apoptosis-related proteins in human bone marrow cells J Immunol Meth 2000 245: 91 94
Philpott NJ, Turner AJ, Scopes J et al. The use of 7-amino actinomycin D in identifying apoptosis: simplicity of use and broad spectrum of application compared with other techniques Blood 1996 87: 2244 2251
Anthony RS, McKelvie ND, Cunningham AJ et al. Flow cytometry using annexin V can detect early apoptosis in peripheral blood precursor cell harvests from patients with leukaemia and lymphoma Bone Marrow Transplant 1998 21: 441 446
Herault O, Colombat P, Domenech J . A rapid single laser flow cytometric method for discrimination of early apoptotic cells in a heterogenous cell population Br J Haematol 1998 104: 530 537
Philpott NJ, Scopes J, Marsh JCW et al. Increased apoptosis in aplastic anemia bone marrow progenitor cells: possible pathophysiologic significance Exp Hematol 1995 23: 1642 1648
Keeney M, Sutherland D . Precursor cell enumeration by flow cytometry: current concepts and recent developments in CD34+ cell enumeration Cytotherapy 2000 2: 395 402
Rowley SD, Feng Z, Yadock D et al. Post-thaw removal of DMSO does not completely abrogate infusional toxicity or the need for pre-infusion histamine blockage Cytotherapy 1999 1: 439 446
Acknowledgements
This work was supported by grants from the Norwegian Cancer Society.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abrahamsen, J., Bakken, A., Bruserud, Ø. et al. Flow cytometric measurement of apoptosis and necrosis in cryopreserved PBPC concentrates from patients with malignant diseases. Bone Marrow Transplant 29, 165–171 (2002). https://doi.org/10.1038/sj.bmt.1703346
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703346
Keywords
This article is cited by
-
Best practices for cryopreserving, thawing, recovering, and assessing cells
In Vitro Cellular & Developmental Biology - Animal (2017)
-
Specific cellular signal-transduction responses to in vivo combination therapy with ATRA, valproic acid and theophylline in acute myeloid leukemia
Blood Cancer Journal (2011)
-
Genes of cell-cell interactions, chemotherapy detoxification and apoptosis are induced during chemotherapy of acute myeloid leukemia
BMC Cancer (2009)