Abstract
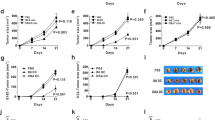
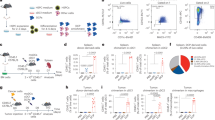
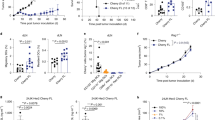
Dendritic cell (DC) vaccination represents an interesting immunotherapeutic option in the treatment of several malignancies. In multiple myeloma (MM) patients, vaccination with autologous idiotype (Id) protein-pulsed DC is feasible, but their antitumoral effectiveness was rather limited. To improve the therapeutic potential of DC therapy, we studied the immunological effects of syngeneic peripheral blood stem cell transplantation (PBSCT) given in conjunction with Id-loaded DC. Balb/c mice were inoculated i.p. with 5 × 105 of HOPC myeloma cells (Balb/c origin). Animals were immunized with three injections of 5 × 105 DC pulsed with the IgG2aHOPC or with a control immunoglobulin (Igcontrol). Some experimental groups of myeloma-bearing animals received total body irradiation (7.5 Gy) and a subsequent transplant of 2 × 107syngeneic peripheral blood progenitor cells (PBPC) followed by DC therapy beginning at day 10 post transplant. Animals receiving DC therapy or syngeneic PBPCT alone neither induce long-term survival nor tumor-specific CTL reactivity in vitro. In marked contrast, combination of syngeneic PBPC transplantation and subsequent DC therapy resulted in 78% survival after a follow-up of 180 days. In addition, this treatment modality conferred a generation of Id peptide-specific CD8-mediated T cell reactivity. These data provide a rationale for DC-based vaccination in multiple myeloma patients administered post syngeneic transplantation.
Bone Marrow Transplantation (2002) 29, 213–221. doi:10.1038/sj.bmt.1703327
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bataille R, Harousseau JL . Multiple myeloma New Engl J Med 1997 336: 1657 1664
Barlogie B, Jaganath S, Vesole D, Tricot G . Autologous and allogeneic transplants for multiple myeloma Semin Hematol 1995 32: 31 44
Attal M, Harousseau JL, Stoppa AM et al. A prospective, randomised trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma New Engl J Med 1996 335: 91 97
Cavo M, Bandini G, Benni M et al. High-dose busulfan and cyclophosphamide are an effective conditioning regimen for allogeneic bone marrow transplantation in chemosensitive multiple myeloma Bone Marrow Transplant 1998 22: 27 32
Sirisinha S, Eisen HN . Autoimmune-like antibodies to the ligand-binding sites of myeloma proteins Proc Natl Acad Sci USA 1971 68: 3130 3135
Lynch RG, Graff RJ, Sirisinha S et al. Myeloma proteins as tumor-specific transplantation antigens Proc Natl Acad Sci USA 1972 69: 1540 1544
Campbell MJ, Carroll W, Kon S et al. Idiotype vaccination against murine B cell lymphoma: humoral and cellular responses elicited by tumor-derived immunoglobulin M and its molecular subunits J Immunol 1987 139: 2825 2833
Kwak LW, Campbell MJ, Zelenetz AD, Levy R . Transfer of specific immunity to B-cell lymphoma with syngeneic bone marrow in mice: a strategy for using autologous marrow as an anti-tumor therapy Blood 1991 78: 2768 2772
Bogen B, Munthe L, Sollien A et al. Naive CD4+ T cells confer idiotype-specific tumor resistance in the absence of antibodies Eur J Immunol 1995 25: 3079 3086
Lauritzsen GF, Weiss S, Dembic Z, Bogen B . Naive idiotype-specific CD4+ T cells and immunosurveillance of B-cell tumors Proc Natl Acad Sci USA 1994 91: 5700 5704
Lauritzsen GF, Bogen B . The role of idiotype-specific, CD4+ T cells in tumor resistance against major histocompatibility complex class II molecule negative plasmacytoma cells Cell Immunol 1993 148: 177 188
Lauritzsen GF, Weiss S, Bogen B . Anti-tumor activity of idiotype-specific, MHC-restricted Th1 and Th2 clones in vitro and in vivo Scand J Immunol 1993 7: 77 85
Porgador A, Gilboa E . Bone marrow-generated dendritic cells pulsed with a class I-restricted peptide are potent inducers of cytotoxic T lymphocytes J Exp Med 1995 182: 255 260
Celluzzi CM, Mayordomo JI, Storkus WJ et al. Peptide-pulsed dendritic cells induce antigen-specific, CTL-mediated protective tumor immunity J Exp Med 1996 183: 283 287
Mayordomo JI, Zorina T, Storkus WJ et al. Bone marrow-derived dendritic cells pulsed with synthetic tumor peptides elicit protective and therapeutic antitumor immunity Nat Med 1995 1: 1297 1302
Flamand V, Sornasse T, Thielemans K et al. Murine dendritic cells pulsed in vitro with tumor antigen induce tumor resistance in vivo Eur J Immunol 1994 24: 605 610
Timmerman JM, Levy R . Dendritic cell vaccines for cancer immunotherapy Ann Rev Med 1999 50: 507 529
Greten TF, Jaffee EM . Cancer vaccines J Clin Oncol 1999 17: 1047 1060
Reichardt VL, Okada CY, Liso A et al. Idiotype vaccination using dendritic cells after autologous peripheral blood stem cell transplantation for multiple myeloma – a feasibility study Blood 1999 93: 2411 2419
Okamoto Y, Gotoh Y, Kobayashi Y et al. Detection of interleukin-12 (IL-12)-secreting cells in normal mice with enzyme-linked immunospot (ELISPOT) assay J Immunoassay 2000 21: 25 37
Glass B, Uharek L, Zeis M et al. Allogeneic peripheral blood progenitor cell transplantation in a murine model: evidence for an improved graft-versus-leukemia effect Blood 1997 90: 1694 1700
Bogen B . Peripheral T-cell tolerance as a tumor escape mechanism: deletion of CD4+ T-cells specific for a monoclonal immunoglobulin idiotype secreted by a plasmocytoma Eur J Immunol 1996 26: 2671 2679
Yi Q, Osterborg A, Bergenbrant S et al. Idiotype-reactive T-cell subsets and tumor load in monoclonal gammopathies Blood 1995 86: 3043 3049
Pawelec G, Heinzel S, Kiessling R et al. Escape mechanisms in tumor immunity: a year 2000 update Crit Rev Oncol 2000 2: 97 133
Salazar-Onfray F . Interleukin-10: a cytokine used by tumors to escape immunosurveilance Med Oncol 1999 16: 86 94
Wen YJ, Ling M, Bailey-Wood R, Lim SH . Idiotypic protein-pulsed adherent peripheral blood mononuclear cell-derived dendritic cells prime immune system in multiple myeloma Clin Cancer Res 1998 4: 957 962
Cull G, Durrant L, Stainer C et al. Generation of anti-idiotype immune responses following vaccination with idiotype-protein pulsed dendritic cells in myeloma Br J Haematol 1999 107: 648 655
Lim SH, Bailey-Wood R . Idiotypic protein-pulsed dendritic cell vaccination in multiple myeloma Int J Cancer 1999 83: 215 222
Titzer S, Christensen O, Manzke O et al. Vaccination of multiple myeloma patients with idiotype-pulsed dendritic cells: immunological and clinical aspects Br J Haematol 2000 108: 805 816
Mackall CL, Bare CV, Granger LA et al. Thymic-independent T-cell regeneration occurs via antigen-driven expansion of peripheral T-cells resulting in a repertoire that is limited in diversity and prone to skewing J Immunol 1996 156: 4609 4616
Qin Z, Blankenstein T . CD4+ T cell-mediated tumor rejection involves inhibition of angiogenesis that is dependent on IFN gamma receptor expression by nonhematopoietic cells Immunity 2000 12: 677 686
Zeis M, Steinmann J, Petrela E et al. Transfer of idiotypic protein primed allogeneic marrow grafts elicits potent graft-versus-myeloma effects in mice Bone Marrow Transplant 2001 27: 279 285
Li Y, Bendandi M, Deng Y et al. Tumor-specific recognition of human myeloma cells by idiotype-induced CD8+ T cells Blood 2000 96: 2828 2833
Österborg A, Yi Q, Henriksson L et al. Idiotype immunization combined with granulocyte–macrophage colony-stimulating factor in myeloma patients induced type I, major histocompatibility complex-restricted, CD8- and CD4-specific T-cell responses Blood 1998 91: 2459 2466
Acknowledgements
This work was supported by the Deutsche Krebshilfe (Grant No. 10–1167 and 70–2428-Schm2).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zeis, M., Frenzke, H., Schmitz, N. et al. Idiotype protein-pulsed dendritic cells produce strong anti-myeloma effects after syngeneic stem cell transplantation in mice. Bone Marrow Transplant 29, 213–221 (2002). https://doi.org/10.1038/sj.bmt.1703327
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703327