Abstract
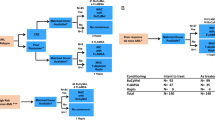
For the treatment of childhood solid tumors, we performed a pilot feasibility study of consecutive high-dose therapies, in which each course was followed by transplantation with granulocyte colony-stimulating factor-mobilized peripheral blood cells which had been separated into CD34-positive and -negative fractions by an Isolex system (Baxter). Positive selection of CD34+ cells has been associated with inevitable cell loss. To overcome this loss, CD34+ cells that had migrated into the negative fraction were saved and used for the first transplant, which was followed by a second transplant after a 3- to 5-month interval. In this phase I feasibility study, the results in six children were evaluated for safety and engraftment. Multi-drug cytoreductive regimens using ranimustine (MCNU), melphalan, thiotepa, carboplatin, cyclophosphamide or VP-16 were comparable between the two transplant procedures in terms of their intensity. The number of CD34+ cells in the ‘CD34(+) fraction’ was 3.31 × 106/kg (0.63–4.3 × 106/kg), while this number in the ‘CD34(−) fraction’ could not be evaluated correctly due their scarcity (<0.1%). the median numbers of infused mnc and cfu-gm were, respectively, 4.2 × 106/kg and 1.75 × 105/kg in the CD34(+) fraction, and 4.8 × 108/kg and 3.35 × 105/kg in the CD34(−) fraction. The number of days required to achieve an ANC >0.5 × 109/l and a platelet count >20 × 109/l and >50 × 109/l were, respectively, 14.5, 15.0 and 19.5 in the first transplant with CD34− cells, and 13.5, 18.0 and 25.0 in the second transplant with CD34+ cells, with no essential difference between the two treatments. Although the small number of patients, the variation in clinical status and treatment, and the short follow-up invalidate any evaluation of the therapeutic benefit of this strategy, the encouraging results support the feasibility of this strategy, which enables an escalation of dose intensity with an improved cost/benefit ratio.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kajiume, T., Kawano, Y., Takaue, Y. et al. New consecutive high-dose chemotherapy modality with fractionated blood stem cell support in the treatment of high-risk pediatric solid tumors: a feasibility study. Bone Marrow Transplant 21, 147–151 (1998). https://doi.org/10.1038/sj.bmt.1701061
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1701061
Keywords
This article is cited by
-
Quimioterapia intensiva con soporte de progenitores hematopoyéticos en neoplasias sólidas no hematológicas de la edad pediátrica: indicaciones y estado actual del tema
Revista de Oncología (2001)
-
Large-scale mobilization and isolation of CD34+ cells from normal donors
Bone Marrow Transplantation (2000)