Abstract
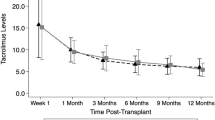
The pharmacokinetics of tacrolimus following its administration as monotherapy or in combination with corticosteroids or methotrexate to 31 BMT patients are presented. All patients received i.v. tacrolimus initially and were subsequently switched to p.o. dosing. Patients received methotrexate by i.v. bolus on post-transplantation days 1, 3, 6 and 11. Patients were started on i.v. corticosteroids beginning on post-transplantation day 7. The noncompartmental pharmacokinetics of tacrolimus based on whole blood concentrations were determined following the i.v. and p.o. doses and were not different at steady-state compared to a single dose. The mean terminal elimination half-life of tacrolimus was 18.2 h following i.v. administration; the total body clearance was 71 ml/h/kg, the volume of distribution was 1.67 l/kg. Co-administration of methylprednisolone or methotrexate did not significantly alter tacrolimus pharmacokinetics. The p.o. bioavailability was 31–49%. Trough blood concentrations (Cmin) at 0 h (pre-dose) and 12 h (post-dose) correlated well to AUC0–12 indicating that, as in solid organ transplantation, Cmin was a good index of drug exposure. Correlation at 0 h (r = 0.92) and at 12 h (r = 0.93) indicate that either time point can be used for therapeutic drug monitoring in patient management.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boswell, G., Bekersky, I., Fay, J. et al. Tacrolimus pharmacokinetics in BMT patients. Bone Marrow Transplant 21, 23–28 (1998). https://doi.org/10.1038/sj.bmt.1701054
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1701054
Keywords
This article is cited by
-
Blood concentration of tacrolimus and age predict tacrolimus-induced left ventricular dysfunction after bone marrow transplantation in adults
Journal of Medical Ultrasonics (2020)
-
Evaluation of Cell-Penetrating Peptides as Versatile, Effective Absorption Enhancers: Relation to Molecular Weight and Inherent Epithelial Drug Permeability
Pharmaceutical Research (2020)
-
Analysis of the variable factors influencing tacrolimus blood concentration during the switch from continuous intravenous infusion to oral administration after allogeneic hematopoietic stem cell transplantation
International Journal of Hematology (2017)
-
Pharmacokinetics, Pharmacodynamics and Pharmacogenomics of Immunosuppressants in Allogeneic Haematopoietic Cell Transplantation: Part I
Clinical Pharmacokinetics (2016)
-
Twice daily i.v. bolus tacrolimus infusion for GVHD prophylaxis in children undergoing stem cell transplantation
Bone Marrow Transplantation (2012)