Abstract
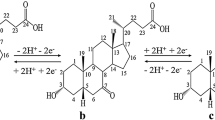
KAWAI1 converted cholic acid to chenodeoxycholic acid (3α : 7α-dihydroxycholanic acid, anthropodeoxycholic acid) by Kishner–Wolff reduction of the derived 3 : 7-dihydroxy-12-keto cholanic acid. Plattner and Heusser2 similarly reduced methyl 3 : 7-di-acetoxy-12-keto cholanate.
Similar content being viewed by others
Article PDF
References
Kawai, S., Hoppe-Seyl Z., 214, 71 (1933).
Plattner, P. A., and Heusser, H., Helv. chim. Acta, 27, 748 (1944).
Fieser, L. F., and Rajogopalan, S., J. Amer. Chem. Soc., 72, 5530 (1950). Fieser, L. F., Rajogopalan, S., Wilson, E., and Tishler, M., J. Amer. Chem. Soc., 73, 4133 (1951).
Huang-Minlon, J. Amer. Chem. Soc., 71, 3301 (1949).
Wootton, I. D. P. (in the press, 1951).
Haslewood, G. A. D., and Wootton, V. M., Biochem. J., 47, 584 (1950).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ANDERSON, I., HASLEWOOD, G., WIGGINS, H. et al. Preparation of Chenodeoxycholic Acid from Cholic Acid. Nature 169, 621 (1952). https://doi.org/10.1038/169621a0
Issue Date:
DOI: https://doi.org/10.1038/169621a0
This article is cited by
-
Chemical and metabolic transformations of selected bile acids
European Journal of Drug Metabolism and Pharmacokinetics (2006)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.