Abstract
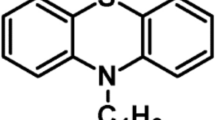
THE term ‘clathrate compound’ has been suggested for that type of molecular complex in which molecules of one compound are held in cavities in the crystal structure of another. Whether a compound can be entrapped in a particular clathrate structure is determined by the physical dimensions of its molecules. Quinol forms clathrate compounds with a number of elements and compounds, and X-ray crystallographic studies of some of these have been made (see, for example, ref. 1). The β-quinol crystal structure contains cavities approximately spherical in shape, the ratio of the number of available cavities to quinol molecules being one to three.
Similar content being viewed by others
Article PDF
References
Palin, D. E., and Powell, H. M., J. Chem. Soc., 815 (1948).
Dryden, J. S., and Meakins, R. J., Nature, [168, 1121 (1951)].
Wesson, L. G., “Tables of Electric Dipole Moments” (Mass. Inst. Tech., 1948).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DRYDEN, J., MEAKINS, R. Dielectric Properties of some Clathrate Compounds of Quinol. Nature 169, 324 (1952). https://doi.org/10.1038/169324a0
Issue Date:
DOI: https://doi.org/10.1038/169324a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.