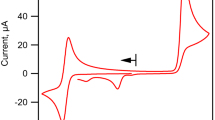
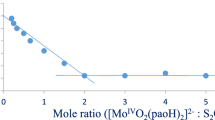
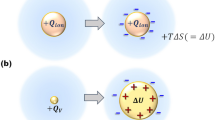
Abstract
IN the course of a spectrophotometric investigation of the hydrolysis of the ferric ion in solutions of ionic strength greater than unity, evidence was obtained that ferric ions entered into complex formation with perchlorate ions. Although it has hitherto been widely assumed that perchlorate anions form no complexes, such interaction might well be expected with the smallest (and most highly charged) cations. Further experiments carried out with approximately 10−4 M ferric perchlorate in 1–7 M perchloric acid solutions in the wave-length range 2600–3600 A. confirmed the preliminary observations and provided quantitative data for the process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Glueckauf, E., Nature, 163, 414 (1949).
Olson, A. R., and Simonson, T. R., J. Chem. Phys., 17, 1322 (1949).
Dodson, R. W., and Silverman, J., Brookhaven National Lab. Quart. Rep. BNL., 64, 82 and 93.
Duke, F. R., and Pinkerton, R. C., J. Amer. Chem. Soc., 73, 3045 (1951).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SUTTON, J. Formation of a Ferric-Perchlorate Ion-Pair Complex. Nature 169, 71–72 (1952). https://doi.org/10.1038/169071b0
Issue Date:
DOI: https://doi.org/10.1038/169071b0
This article is cited by
-
Ferric Perchlorate Complex Ion
Nature (1958)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.