Abstract
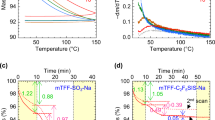
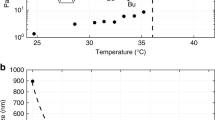
Batchelor and Foster1 found, by examination of the vapour pressure – temperature curves, that dioxan adsorbed on a ferric oxide gel of pore radius ∼ 100 A. melts sharply about 6° C. below the normal melting point of the bulk liquid. During a recent investigation of the sorption of amines by silica gel, a similar effect has been observed with ethylenediamine.
Similar content being viewed by others
Article PDF
References
Trans. Farad. Soc., 40, 301 (1944).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BROWN, M., FOSTER, A. Melting Point of Adsorbed Liquids. Nature 169, 37 (1952). https://doi.org/10.1038/169037b0
Issue Date:
DOI: https://doi.org/10.1038/169037b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.