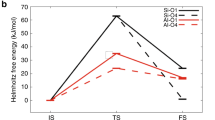
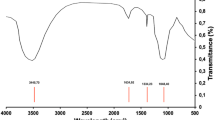
Abstract
IN the course of a study of the water sorption of aluminium soaps, Shreve, Pomeroy and Mysels1 found that pure aluminium laurate which had been dried in vacuo over phosphorus pentoxide at room temperature gave 2·5 ± 0·4 per cent of water when examined with the Karl Fischer reagent. They maintained, consequently, that the reagent gives misleading results because, under the conditions of the determination, soap molecules react with each other, or with free acid, to give water, thus:
Similar content being viewed by others
Article PDF
References
Shreve, Pomeroy and Mysels, J. Phys. Coll. Chem., 51, 963 (1947).
Schulman and McRoberts, Nature, 162, 101 (1948).
Marion and Thomas, J. Colloid Science, 1, 221 (1946).
Bryant, Mitchell, Smith and Ashby, J. Amer. Chem. Soc., 63, 2924 (1941).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PARRY, G., TAYLOR, A. Structure of Aluminium Soaps. Nature 164, 449 (1949). https://doi.org/10.1038/164449a0
Issue Date:
DOI: https://doi.org/10.1038/164449a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.